Thyroid-Stimulating Hormone Inhibits Insulin Receptor Substrate-1 Expression and Tyrosyl Phosphorylation in 3T3-L1 Adipocytes by Increasing NF- κ B DNA-Binding Activity
- PMID: 35320949
- PMCID: PMC8938072
- DOI: 10.1155/2022/7553670
Thyroid-Stimulating Hormone Inhibits Insulin Receptor Substrate-1 Expression and Tyrosyl Phosphorylation in 3T3-L1 Adipocytes by Increasing NF- κ B DNA-Binding Activity
Abstract
Background: Abundant evidence indicates that thyroid-stimulating hormone (TSH) levels are associated with insulin resistance in adipocytes. However, the potential mechanism of the association remains uncertain. The objective of this study was to determine the potential role of TSH in the suppression of insulin receptor substrate-1 (IRS-1) expression and IRS-1 tyrosyl phosphorylation, which might contribute to insulin resistance.
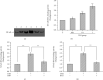
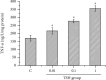
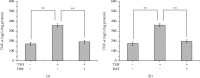
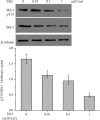
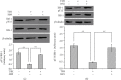
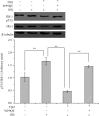
Methods: Mouse 3T3-L1 preadipocytes were differentiated into adipocytes. After treatment with 0.01, 0.1, and 1.0 mIU/ml bovine TSH, the TNF-α concentration in the medium was determined by enzyme-linked immunosorbent assay (ELISA). Nuclear factor-kappa B (NF-κB) DNA-binding activity was quantified by electrophoretic mobility shift assay (EMSA). IRS-1 levels in adipocytes were quantified by Western blotting, and tyrosine phosphorylation was measured by immunoprecipitation.
Results: TSH induced TNF-α secretion in a dose-dependent manner. There was a significant positive correlation between NF-κB DNA-binding activity and TNF-α secretion. This effect and correlation were weakened by BAY 11-7082 (a nuclear NF-κB inhibitor) and H89 (an inhibitor of cyclic adenosine monophosphate- (cAMP-) dependent protein kinase A (PKA)). Treatment of cultured adipocytes with TSH inhibited insulin-stimulated IRS-1 tyrosyl phosphorylation but promoted TSH-dependent secretion of TNF-α and activation of NF-κB DNA-binding activity. The effects of TSH were significantly inhibited by BAY 11-7082 and H89 and were completely blocked by the TNF-α antagonist WP9QY.
Conclusion: TSH inhibited IRS-1 protein expression and tyrosyl phosphorylation in 3T3-L1 adipocytes by stimulating TNF-α production via promotion of NF-κB DNA-binding activity. TSH might play a pivotal role in the development of insulin resistance.
Copyright © 2022 Yajing Zhang and Ling Feng.
Conflict of interest statement
There are no conflicts of interest to declare.
Figures
Similar articles
-
Thyroid-stimulating hormone induces the secretion of tumor necrosis factor-α from 3T3-L1 adipocytes via a protein kinase A-dependent pathway.Exp Clin Endocrinol Diabetes. 2013 Aug;121(8):488-93. doi: 10.1055/s-0033-1347266. Epub 2013 Jul 17. Exp Clin Endocrinol Diabetes. 2013. PMID: 23864495
-
HM-chromanone attenuates TNF-α-mediated inflammation and insulin resistance by controlling JNK activation and NF-κB pathway in 3T3-L1 adipocytes.Eur J Pharmacol. 2022 Apr 15;921:174884. doi: 10.1016/j.ejphar.2022.174884. Epub 2022 Mar 11. Eur J Pharmacol. 2022. PMID: 35288193
-
Tumor necrosis factor-alpha suppresses adipocyte-specific genes and activates expression of preadipocyte genes in 3T3-L1 adipocytes: nuclear factor-kappaB activation by TNF-alpha is obligatory.Diabetes. 2002 May;51(5):1319-36. doi: 10.2337/diabetes.51.5.1319. Diabetes. 2002. PMID: 11978627
-
The mechanisms of carnosic acid attenuates tumor necrosis factor-α-mediated inflammation and insulin resistance in 3T3-L1 adipocytes.Mol Nutr Food Res. 2014 Apr;58(4):654-64. doi: 10.1002/mnfr.201300356. Epub 2013 Nov 11. Mol Nutr Food Res. 2014. PMID: 24668853
-
Effect of NF-κB decoy on insulin resistance of adipocytes from patients with type 2 diabetes mellitus.Diabetes Metab. 2011 Dec;37(6):520-6. doi: 10.1016/j.diabet.2011.04.004. Epub 2011 Jun 12. Diabetes Metab. 2011. PMID: 21664164
Cited by
-
Effects of Anterior Pituitary Adenomas' Hormones on Glucose Metabolism and Its Clinical Implications.Diabetes Metab Syndr Obes. 2023 Feb 13;16:409-424. doi: 10.2147/DMSO.S397445. eCollection 2023. Diabetes Metab Syndr Obes. 2023. PMID: 36816815 Free PMC article. Review.
-
Impaired sensitivity to thyroid hormones is associated with increased body fat mass/muscle mass ratio (F/M) in the euthyroid population.Diabetol Metab Syndr. 2025 Apr 15;17(1):128. doi: 10.1186/s13098-025-01693-w. Diabetol Metab Syndr. 2025. PMID: 40234912 Free PMC article.
-
Increased thyroid stimulating hormone (TSH) as a possible risk factor for atherosclerosis in subclinical hypothyroidism.Thyroid Res. 2024 Jun 17;17(1):13. doi: 10.1186/s13044-024-00199-3. Thyroid Res. 2024. PMID: 38880884 Free PMC article. Review.
-
Giardia duodenalis enolase is secreted as monomer during trophozoite-epithelial cell interactions, activates plasminogen and induces necroptotic damage.Front Cell Infect Microbiol. 2022 Aug 25;12:928687. doi: 10.3389/fcimb.2022.928687. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 36093180 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

