A toolkit for recombinant production of seven human EGF family growth factors in active conformation
- PMID: 35322149
- PMCID: PMC8943033
- DOI: 10.1038/s41598-022-09060-9
A toolkit for recombinant production of seven human EGF family growth factors in active conformation
Abstract
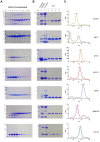
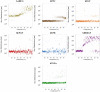
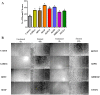
Epidermal growth factors (EGF) play a wide range of roles in embryogenesis, skin development, immune response homeostasis. They are involved in several pathologies as well, including several cancer types, psoriasis, chronic pain and chronic kidney disease. All members share the structural EGF domain, which is responsible for receptor interaction, thereby initiating transduction of signals. EGF growth factors have intense use in fundamental research and high potential for biotechnological applications. However, due to their structural organization with three disulfide bonds, recombinant production of these factors in prokaryotic systems is not straightforward. A significant fraction usually forms inclusion bodies. For the fraction remaining soluble, misfolding and incomplete disulfide bond formation may affect the amount of active factor in solution, which can compromise experimental conclusions and biotechnological applications. In this work, we describe a reliable procedure to produce seven human growth factors of the EGF family in Escherichia coli. Biophysical and stability analyses using limited proteolysis, light scattering, circular dichroism and nanoDSF show that the recombinant factors present folded and stable conformation. Cell proliferation and scratch healing assays confirmed that the recombinant factors are highly active at concentrations as low as 5 ng/ml.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





References
-
- Yang CC, Chang KW. Eicosanoids and HB-EGF/EGFR in cancer. Cancer Metastasis Rev. 2018;37:385–395. - PubMed
-
- Zhao J, et al. Betacellulin enhances ovarian cancer cell migration by up-regulating Connexin43 via MEK-ERK signaling. Cell. Signal. 2020;65:109439. - PubMed
-
- Carvalho S, et al. An antibody to amphiregulin, an abundant growth factor in patients’ fluids, inhibits ovarian tumors. Oncogene. 2016;35:438–447. - PubMed
-
- Liu Y. Epidermal growth factor on the healing of human subacute tympanic membrane perforation. Am. J. Otolaryngol. 2020;41:102399. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

