The Real-World Experience of the Biosimilar (Grastofil®) to the Reference Biologic (Neupogen®) in Breast Cancer and Lymphoma: A Canadian Single-Centre Retrospective Study
- PMID: 35323315
- PMCID: PMC8947031
- DOI: 10.3390/curroncol29030115
The Real-World Experience of the Biosimilar (Grastofil®) to the Reference Biologic (Neupogen®) in Breast Cancer and Lymphoma: A Canadian Single-Centre Retrospective Study
Abstract
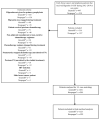
Febrile neutropenia (FN) is a common side effect of cytotoxic chemotherapy that may result in poor treatment outcomes. The short acting granulocyte colony stimulating factors (G-CSF) act to stimulate granulocytes to increase production of white blood cells. The filgrastim biosimilar is useful, as it may provide a cheaper and equally effective treatment to FN. This study explored the usage of the filgrastim biosimilar (Grastofil®) and the reference biologic (Neupogen®) in breast cancer and lymphoma patients. A retrospective chart review of patients receiving Grastofil® from January 2017 to June 2019 or Neupogen® for primary prophylaxis of FN from January 2013 to December 2017 was conducted. The endpoints included the incidence of FN and the occurrence of dose reduction (DR) and dose delay (DD). One hundred and fifty-three Grastofil® patients were matched to 153 Neupogen® patients. This cohort was further split into breast cancer (n = 275) and non-Hodgkin's lymphoma (n = 31) cohorts. After adjusting for chemotherapy cycles, the biosimilar filgrastim was non-inferior to the reference biologic based on FN incidence in addition to related outcomes including DR and DD.
Keywords: Neupogen® and Grastofil®; biosimilar; breast cancer; febrile neutropenia; lymphoma; primary prophylaxis; retrospective study.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Real World Clinical Experience of Biosimilar G-CSF (Grastofil) for Autologous Peripheral Blood Stem Cell Mobilization: Single Center Experience in Canada Following Early Adoption.Curr Oncol. 2021 Apr 22;28(3):1571-1580. doi: 10.3390/curroncol28030148. Curr Oncol. 2021. PMID: 33922026 Free PMC article.
-
A retrospective review of the real-world experience of the Pegfilgrastim biosimilar (Lapelga®) to the reference biologic (Neulasta®).J Oncol Pharm Pract. 2022 Jan;28(1):5-16. doi: 10.1177/1078155220974085. Epub 2020 Nov 20. J Oncol Pharm Pract. 2022. PMID: 33215563 Free PMC article.
-
Safety and efficacy of alternating treatment with EP2006, a filgrastim biosimilar, and reference filgrastim: a phase III, randomised, double-blind clinical study in the prevention of severe neutropenia in patients with breast cancer receiving myelosuppressive chemotherapy.Ann Oncol. 2018 Jan 1;29(1):244-249. doi: 10.1093/annonc/mdx638. Ann Oncol. 2018. PMID: 29091995 Clinical Trial.
-
Biosimilar Pegfilgrastim: Improving Access and Optimising Practice to Supportive Care that Enables Cure.BioDrugs. 2020 Jun;34(3):255-263. doi: 10.1007/s40259-020-00411-4. BioDrugs. 2020. PMID: 32232676 Free PMC article. Review.
-
Tbo-Filgrastim: A Review in Neutropenic Conditions.BioDrugs. 2016 Apr;30(2):153-60. doi: 10.1007/s40259-016-0172-7. BioDrugs. 2016. PMID: 27023705 Review.
References
-
- Weycker D., Barron R., Edelsberg J., Kartashov A., Legg J., Glass A.G. Risk and Consequences of Chemotherapy-Induced Neutropenic Complications in Patients Receiving Daily Filgrastim: The Importance of Duration of Prophylaxis. BMC Health Serv. Res. 2014;14:189. doi: 10.1186/1472-6963-14-189. - DOI - PMC - PubMed
-
- Smith T.J., Bohlke K., Lyman G.H., Carson K.R., Crawford J., Cross S.J., Goldberg J.M., Khatcheressian J.L., Leighl N.B., Perkins C.L., et al. Recommendations for the Use of WBC Growth Factors: American Society of Clinical Oncology Clinical Practice Guideline Update. J. Clin. Oncol. 2015;33:3199–3212. doi: 10.1200/JCO.2015.62.3488. - DOI - PubMed
-
- Lyman G.H., Dale D.C., Culakova E., Poniewierski M.S., Wolff D.A., Kuderer N.M., Huang M., Crawford J. The Impact of the Granulocyte Colony-Stimulating Factor on Chemotherapy Dose Intensity and Cancer Survival: A Systematic Review and Meta-Analysisof Randomized Controlled Trials. Ann. Oncol. 2013;24:2475–2484. doi: 10.1093/annonc/mdt226. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous