Novel Strategies to Inhibit Pertussis Toxin
- PMID: 35324684
- PMCID: PMC8951090
- DOI: 10.3390/toxins14030187
Novel Strategies to Inhibit Pertussis Toxin
Abstract
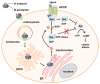
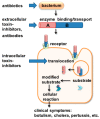
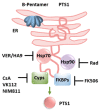
Pertussis, also known as whooping cough, is a respiratory disease caused by infection with Bordetella pertussis, which releases several virulence factors, including the AB-type pertussis toxin (PT). The characteristic symptom is severe, long-lasting paroxysmal coughing. Especially in newborns and infants, pertussis symptoms, such as leukocytosis, can become life-threatening. Despite an available vaccination, increasing case numbers have been reported worldwide, including Western countries such as Germany and the USA. Antibiotic treatment is available and important to prevent further transmission. However, antibiotics only reduce symptoms if administered in early stages, which rarely occurs due to a late diagnosis. Thus, no causative treatments against symptoms of whooping cough are currently available. The AB-type protein toxin PT is a main virulence factor and consists of a binding subunit that facilitates transport of an enzyme subunit into the cytosol of target cells. There, the enzyme subunit ADP-ribosylates inhibitory α-subunits of G-protein coupled receptors resulting in disturbed cAMP signaling. As an important virulence factor associated with severe symptoms, such as leukocytosis, and poor outcomes, PT represents an attractive drug target to develop novel therapeutic strategies. In this review, chaperone inhibitors, human peptides, small molecule inhibitors, and humanized antibodies are discussed as novel strategies to inhibit PT.
Keywords: ADP-ribosylation inhibitor; chaperones; human defensins; humanized antibodies; novel inhibitors; pertussis toxin.
Conflict of interest statement
The author declares no conflict of interest.
Figures
Similar articles
-
Human Peptides α-Defensin-1 and -5 Inhibit Pertussis Toxin.Toxins (Basel). 2021 Jul 11;13(7):480. doi: 10.3390/toxins13070480. Toxins (Basel). 2021. PMID: 34357952 Free PMC article.
-
Alpha-1 antitrypsin inhibits pertussis toxin.J Biol Chem. 2024 Dec;300(12):107950. doi: 10.1016/j.jbc.2024.107950. Epub 2024 Oct 30. J Biol Chem. 2024. PMID: 39481600 Free PMC article.
-
Pharmacological targeting of host chaperones protects from pertussis toxin in vitro and in vivo.Sci Rep. 2021 Mar 8;11(1):5429. doi: 10.1038/s41598-021-84817-2. Sci Rep. 2021. PMID: 33686161 Free PMC article.
-
The concept of pertussis as a toxin-mediated disease.Pediatr Infect Dis. 1984 Sep-Oct;3(5):467-86. doi: 10.1097/00006454-198409000-00019. Pediatr Infect Dis. 1984. PMID: 6093069 Review.
-
Investigating pertussis toxin and its impact on vaccination.Future Microbiol. 2015;10(2):241-54. doi: 10.2217/fmb.14.123. Future Microbiol. 2015. PMID: 25689536 Review.
Cited by
-
Development and validation of a diagnostic prediction model for children with pertussis.Sci Rep. 2024 Jul 26;14(1):17154. doi: 10.1038/s41598-024-65856-x. Sci Rep. 2024. PMID: 39060316 Free PMC article.
-
Inhibition of Pertussis Toxin by Human α-Defensins-1 and -5: Differential Mechanisms of Action.Int J Mol Sci. 2023 Jun 23;24(13):10557. doi: 10.3390/ijms241310557. Int J Mol Sci. 2023. PMID: 37445740 Free PMC article.
-
Domperidone Inhibits Clostridium botulinum C2 Toxin and Bordetella pertussis Toxin.Toxins (Basel). 2023 Jun 25;15(7):412. doi: 10.3390/toxins15070412. Toxins (Basel). 2023. PMID: 37505681 Free PMC article.
-
The antiarrhythmic drugs amiodarone and dronedarone inhibit intoxication of cells with pertussis toxin.Naunyn Schmiedebergs Arch Pharmacol. 2024 Dec;397(12):9991-10003. doi: 10.1007/s00210-024-03247-9. Epub 2024 Jul 3. Naunyn Schmiedebergs Arch Pharmacol. 2024. PMID: 38958734 Free PMC article.
-
Exploiting subtractive genomics to identify novel drug targets and new immunogenic candidates against Bordetella pertussis: an in silico study.Front Bioinform. 2025 May 13;5:1570054. doi: 10.3389/fbinf.2025.1570054. eCollection 2025. Front Bioinform. 2025. PMID: 40433465 Free PMC article.
References
-
- Robert-Koch-Institut Infektionsepidemiologisches Jahrbuch Meldepflichtiger Krankheiten für 2018. Robert Koch Institute; Berlin, Germany: 2018. 247p
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

