Selection, Characterization, and Optimization of DNA Aptamers against Challenging Marine Biotoxin Gymnodimine-A for Biosensing Application
- PMID: 35324692
- PMCID: PMC8949142
- DOI: 10.3390/toxins14030195
Selection, Characterization, and Optimization of DNA Aptamers against Challenging Marine Biotoxin Gymnodimine-A for Biosensing Application
Abstract
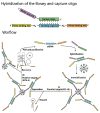
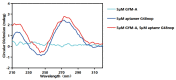
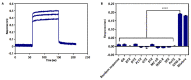
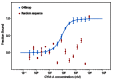
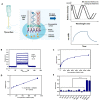
Gymnodimines (GYMs), belonging to cyclic imines (CIs), are characterized as fast-acting toxins, and may pose potential risks to human health and the aquaculture industry through the contamination of sea food. The existing detection methods of GYMs have certain defects in practice, such as ethical problems or the requirement of complicated equipment. As novel molecular recognition elements, aptamers have been applied in many areas, including the detection of marine biotoxins. However, GYMs are liposoluble molecules with low molecular weight and limited numbers of chemical groups, which are considered as "challenging" targets for aptamers selection. In this study, Capture-SELEX was used as the main strategy in screening aptamers targeting gymnodimine-A (GYM-A), and an aptamer named G48nop, with the highest KD value of 95.30 nM, was successfully obtained by screening and optimization. G48nop showed high specificity towards GYM-A. Based on this, a novel aptasensor based on biolayer interferometry (BLI) technology was established in detecting GYM-A. This aptasensor showed a detection range from 55 to 1400 nM (linear range from 55 to 875 nM) and a limit of detection (LOD) of 6.21 nM. Spiking experiments in real samples indicated the recovery rate of this aptasensor, ranging from 96.65% to 109.67%. This is the first study to report an aptamer with high affinity and specificity for the challenging marine biotoxin GYM-A, and the new established aptasensor may be used as a reliable and efficient tool for the detection and monitoring of GYMs in the future.
Keywords: aptamer; aptasensor; biolayer interferometry; gymnodimine-A.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

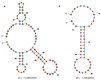
Similar articles
-
Brevetoxin Aptamer Selection and Biolayer Interferometry Biosensor Application.Toxins (Basel). 2024 Sep 24;16(10):411. doi: 10.3390/toxins16100411. Toxins (Basel). 2024. PMID: 39453187 Free PMC article.
-
Gonyautoxin 1/4 aptamers with high-affinity and high-specificity: From efficient selection to aptasensor application.Biosens Bioelectron. 2016 May 15;79:938-44. doi: 10.1016/j.bios.2016.01.032. Epub 2016 Jan 12. Biosens Bioelectron. 2016. PMID: 26802576
-
A Novel SELEX Based on Immobilizing Libraries Enables Screening of Saxitoxin Aptamers for BLI Aptasensor Applications.Toxins (Basel). 2022 Mar 21;14(3):228. doi: 10.3390/toxins14030228. Toxins (Basel). 2022. PMID: 35324725 Free PMC article.
-
Aptamers and Aptasensors for Highly Specific Recognition and Sensitive Detection of Marine Biotoxins: Recent Advances and Perspectives.Toxins (Basel). 2018 Oct 25;10(11):427. doi: 10.3390/toxins10110427. Toxins (Basel). 2018. PMID: 30366456 Free PMC article. Review.
-
Marine Toxins Detection by Biosensors Based on Aptamers.Toxins (Basel). 2019 Dec 18;12(1):1. doi: 10.3390/toxins12010001. Toxins (Basel). 2019. PMID: 31861315 Free PMC article. Review.
Cited by
-
The Potential of Single-Chain Variable Fragment Antibody: Role in Future Therapeutic and Diagnostic Biologics.J Immunol Res. 2024 Aug 9;2024:1804038. doi: 10.1155/2024/1804038. eCollection 2024. J Immunol Res. 2024. PMID: 39156005 Free PMC article. Review.
-
High Affinity Aptamers and Their Specificity for Azaspiracid-2 Using Capture-SELEX.Mar Drugs. 2025 Apr 25;23(5):183. doi: 10.3390/md23050183. Mar Drugs. 2025. PMID: 40422773 Free PMC article.
-
Capture-SELEX: Selection Strategy, Aptamer Identification, and Biosensing Application.Biosensors (Basel). 2022 Dec 7;12(12):1142. doi: 10.3390/bios12121142. Biosensors (Basel). 2022. PMID: 36551109 Free PMC article. Review.
-
Perspectives in Aptasensor-Based Portable Detection for Biotoxins.Molecules. 2024 Oct 15;29(20):4891. doi: 10.3390/molecules29204891. Molecules. 2024. PMID: 39459259 Free PMC article. Review.
-
Programmed Aptamer Screening, Characterization, and Rapid Detection for α-Conotoxin MI.Toxins (Basel). 2022 Oct 14;14(10):706. doi: 10.3390/toxins14100706. Toxins (Basel). 2022. PMID: 36287974 Free PMC article.
References
-
- Takahashi E., Yu Q., Eaglesham G., Connell D.W., McBroom J., Costanzo S., Shaw G.R. Occurrence and seasonal variations of algal toxins in water, phytoplankton and shellfish from North Stradbroke Island, Queensland, Australia. Mar. Environ. Res. 2007;64:429–442. doi: 10.1016/j.marenvres.2007.03.005. - DOI - PubMed
-
- Stirling D.J. Survey of historical New Zealand shellfish samples for accumulation of gymnodimine. N. Z. J. Mar. Freshw. Res. 2001;35:851–857. doi: 10.1080/00288330.2001.9517047. - DOI
-
- Seki T., Satake M., Mackenzie L., Kaspar H.F., Yasumoto T. Gymnodimine, a new marine toxin of unprecedented structure isolated from New Zealand oysters and the dinoflagellate, Gymnodinium sp. Tetrahedron Lett. 1995;36:7093–7096. doi: 10.1016/0040-4039(95)01434-J. - DOI
-
- Salgado P., Riobó P., Rodríguez F., Franco J.M., Bravo I. Differences in the toxin profiles of Alexandrium ostenfeldii (Dinophyceae) strains isolated from different geographic origins: Evidence of paralytic toxin, spirolide, and gymnodimine. Toxicon. 2015;103:85–98. doi: 10.1016/j.toxicon.2015.06.015. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

