STxB as an Antigen Delivery Tool for Mucosal Vaccination
- PMID: 35324699
- PMCID: PMC8948715
- DOI: 10.3390/toxins14030202
STxB as an Antigen Delivery Tool for Mucosal Vaccination
Abstract
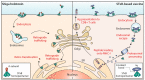
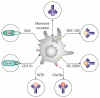
Immunotherapy against cancer and infectious disease holds the promise of high efficacy with minor side effects. Mucosal vaccines to protect against tumors or infections disease agents that affect the upper airways or the lung are still lacking, however. One mucosal vaccine candidate is the B-subunit of Shiga toxin, STxB. In this review, we compare STxB to other immunotherapy vectors. STxB is a non-toxic protein that binds to a glycosylated lipid, termed globotriaosylceramide (Gb3), which is preferentially expressed by dendritic cells. We review the use of STxB for the cross-presentation of tumor or viral antigens in a MHC class I-restricted manner to induce humoral immunity against these antigens in addition to polyfunctional and persistent CD4+ and CD8+ T lymphocytes capable of protecting against viral infection or tumor growth. Other literature will be summarized that documents a powerful induction of mucosal IgA and resident memory CD8+ T cells against mucosal tumors specifically when STxB-antigen conjugates are administered via the nasal route. It will also be pointed out how STxB-based vaccines have been shown in preclinical cancer models to synergize with other therapeutic modalities (immune checkpoint inhibitors, anti-angiogenic therapy, radiotherapy). Finally, we will discuss how molecular aspects such as low immunogenicity, cross-species conservation of Gb3 expression, and lack of toxicity contribute to the competitive positioning of STxB among the different DC targeting approaches. STxB thereby appears as an original and innovative tool for the development of mucosal vaccines in infectious diseases and cancer.
Keywords: GL-Lect; TRM; chemotherapy; cross-presentation; cytotoxic CD8+ T lymphocyte; endosomal escape; glycolipid-lectin; immune checkpoint; radiotherapy; tissue resident memory T cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Retrograde transport is not required for cytosolic translocation of the B-subunit of Shiga toxin.J Cell Sci. 2015 Jul 1;128(13):2373-87. doi: 10.1242/jcs.169383. Epub 2015 May 14. J Cell Sci. 2015. PMID: 25977475
-
The Shiga toxin B-subunit targets antigen in vivo to dendritic cells and elicits anti-tumor immunity.Eur J Immunol. 2006 May;36(5):1124-35. doi: 10.1002/eji.200535443. Eur J Immunol. 2006. PMID: 16568496
-
The B subunit of Shiga toxin coupled to full-size antigenic protein elicits humoral and cell-mediated immune responses associated with a Th1-dominant polarization.Int Immunol. 2003 Oct;15(10):1161-71. doi: 10.1093/intimm/dxg118. Int Immunol. 2003. PMID: 13679386
-
Resident memory T cells, critical components in tumor immunology.J Immunother Cancer. 2018 Sep 4;6(1):87. doi: 10.1186/s40425-018-0399-6. J Immunother Cancer. 2018. PMID: 30180905 Free PMC article. Review.
-
Therapeutic vaccination with tumor cells that engage CD137.J Mol Med (Berl). 2003 Feb;81(2):71-86. doi: 10.1007/s00109-002-0413-8. Epub 2003 Feb 8. J Mol Med (Berl). 2003. PMID: 12601523 Review.
Cited by
-
Dimeric Lectin Chimeras as Novel Candidates for Gb3-Mediated Transcytotic Drug Delivery through Cellular Barriers.Pharmaceutics. 2023 Jan 9;15(1):225. doi: 10.3390/pharmaceutics15010225. Pharmaceutics. 2023. PMID: 36678854 Free PMC article.
-
CXCR6 expressing T cells: Functions and role in the control of tumors.Front Immunol. 2022 Oct 12;13:1022136. doi: 10.3389/fimmu.2022.1022136. eCollection 2022. Front Immunol. 2022. PMID: 36311728 Free PMC article. Review.
-
Engineered Synthetic STxB for Enhanced Cytosolic Delivery.Cells. 2023 Apr 30;12(9):1291. doi: 10.3390/cells12091291. Cells. 2023. PMID: 37174690 Free PMC article.
-
Immunogenicity of inactivated Escherichia coli O157:H7 with Stx2B microparticle in mice.Iran J Basic Med Sci. 2022 Sep;25(9):1069-1076. doi: 10.22038/IJBMS.2022.63775.14053. Iran J Basic Med Sci. 2022. PMID: 36246065 Free PMC article.
-
Martini 3 Coarse-Grained Force Field for Carbohydrates.J Chem Theory Comput. 2022 Dec 13;18(12):7555-7569. doi: 10.1021/acs.jctc.2c00757. Epub 2022 Nov 7. J Chem Theory Comput. 2022. PMID: 36342474 Free PMC article.
References
-
- Jacewicz M., Clausen H., Nudelman E., Donohue-Rolfe A., Keusch G.T. Pathogenesis of shigella diarrhea. XI. Isolation of a shigella toxin-binding glycolipid from rabbit jejunum and HeLa cells and its identification as globotriaosylceramide. J. Exp. Med. 1986;163:1391–1404. doi: 10.1084/jem.163.6.1391. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

