Exosomes in the Pathogenesis, Progression, and Treatment of Osteoarthritis
- PMID: 35324788
- PMCID: PMC8945849
- DOI: 10.3390/bioengineering9030099
Exosomes in the Pathogenesis, Progression, and Treatment of Osteoarthritis
Abstract
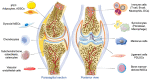
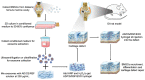
Osteoarthritis (OA) is a prevalent and debilitating age-related joint disease characterized by articular cartilage degeneration, synovial membrane inflammation, osteophyte formation, as well as subchondral bone sclerosis. OA drugs at present are mainly palliative and do not halt or reverse disease progression. Currently, no disease-modifying OA drugs (DMOADs) are available and total joint arthroplasty remains a last resort. Therefore, there is an urgent need for the development of efficacious treatments for OA management. Among all novel pharmaco-therapeutical options, exosome-based therapeutic strategies are highly promising. Exosome cargoes, which include proteins, lipids, cytokines, and various RNA subtypes, are potentially capable of regulating intercellular communications and gene expression in target cells and tissues involved in OA development. With extensive research in recent years, exosomes in OA studies are no longer limited to classic, mesenchymal stem cell (MSC)-derived vesicles. New origins, structures, and functions of exosomes are constantly being discovered and investigated. This review systematically summarizes the non-classic origins, biosynthesis, and extraction of exosomes, describes modification and delivery techniques, explores their role in OA pathogenesis and progression, and discusses their therapeutic potential and hurdles to overcome in OA treatment.
Keywords: cartilage injury; chondrocyte; exosome; extracellular vesicle; osteoarthritis; regenerative medicine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Exosomes in intercellular communication and implications for osteoarthritis.Rheumatology (Oxford). 2020 Jan 1;59(1):57-68. doi: 10.1093/rheumatology/kez462. Rheumatology (Oxford). 2020. PMID: 31628481 Review.
-
Potential of Exosomes as Cell-Free Therapy in Articular Cartilage Regeneration: A Review.Int J Nanomedicine. 2021 Oct 1;16:6749-6781. doi: 10.2147/IJN.S327059. eCollection 2021. Int J Nanomedicine. 2021. PMID: 34621125 Free PMC article. Review.
-
Exosome-Mediated Drug Delivery for Cell-Free Therapy of Osteoarthritis.Curr Med Chem. 2021;28(31):6458-6483. doi: 10.2174/0929867327666201118161232. Curr Med Chem. 2021. PMID: 33213308 Review.
-
Osteoarthritic Subchondral Bone Release Exosomes That Promote Cartilage Degeneration.Cells. 2021 Jan 28;10(2):251. doi: 10.3390/cells10020251. Cells. 2021. PMID: 33525381 Free PMC article.
-
The dual character of exosomes in osteoarthritis: Antagonists and therapeutic agents.Acta Biomater. 2020 Mar 15;105:15-25. doi: 10.1016/j.actbio.2020.01.040. Epub 2020 Feb 6. Acta Biomater. 2020. PMID: 32006653 Review.
Cited by
-
Stem Cell Exosomes for Osteoarthritis in Veterinary Medicine.Stem Cells Int. 2025 Jul 16;2025:4888569. doi: 10.1155/sci/4888569. eCollection 2025. Stem Cells Int. 2025. PMID: 40704321 Free PMC article. Review.
-
Causal association of menstrual reproductive factors on the risk of osteoarthritis: A univariate and multivariate Mendelian randomization study.PLoS One. 2024 Aug 30;19(8):e0307958. doi: 10.1371/journal.pone.0307958. eCollection 2024. PLoS One. 2024. PMID: 39213290 Free PMC article.
-
Mesenchymal Stromal Cells as a Driver of Inflammaging.Int J Mol Sci. 2023 Mar 28;24(7):6372. doi: 10.3390/ijms24076372. Int J Mol Sci. 2023. PMID: 37047346 Free PMC article. Review.
-
Current Non-Surgical Curative Regenerative Therapies for Knee Osteoarthritis.Stem Cell Rev Rep. 2024 Nov;20(8):2104-2123. doi: 10.1007/s12015-024-10768-6. Epub 2024 Aug 15. Stem Cell Rev Rep. 2024. PMID: 39145857 Review.
-
Exosomes derived from miR-146a-overexpressing fibroblast-like synoviocytes in cartilage degradation and macrophage M1 polarization: a novel protective agent for osteoarthritis?Front Immunol. 2024 May 23;15:1361606. doi: 10.3389/fimmu.2024.1361606. eCollection 2024. Front Immunol. 2024. PMID: 38846937 Free PMC article.
References
-
- Favero M., El-Hadi H., Belluzzi E., Granzotto M., Porzionato A., Sarasin G., Rambaldo A., Iacobellis C., Cigolotti A., Fontanella C.G., et al. Infrapatellar fat pad features in osteoarthritis: A histopathological and molecular study. Rheumatology (Oxford) 2017;56:1784–1793. doi: 10.1093/rheumatology/kex287. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

