TERT promoter C228T mutation in neural progenitors confers growth advantage following telomere shortening in vivo
- PMID: 35325218
- PMCID: PMC9713509
- DOI: 10.1093/neuonc/noac080
TERT promoter C228T mutation in neural progenitors confers growth advantage following telomere shortening in vivo
Erratum in
-
Corrigendum to: TERT promoter C228T mutation in neural progenitors confers growth advantage following telomere shortening in vivo.Neuro Oncol. 2022 Nov 2;24(11):2008. doi: 10.1093/neuonc/noac178. Neuro Oncol. 2022. PMID: 35876482 Free PMC article. No abstract available.
Abstract
Background: Heterozygous TERT (telomerase reverse transcriptase) promoter mutations (TPMs) facilitate TERT expression and are the most frequent mutation in glioblastoma (GBM). A recent analysis revealed this mutation is one of the earliest events in gliomagenesis. However, no appropriate human models have been engineered to study the role of this mutation in the initiation of these tumors.
Method: We established GBM models by introducing the heterozygous TPM in human induced pluripotent stem cells (hiPSCs) using a two-step targeting approach in the context of GBM genetic alterations, CDKN2A/B and PTEN deletion, and EGFRvIII overexpression. The impact of the mutation was evaluated through the in vivo passage and in vitro experiment and analysis.
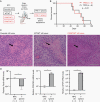
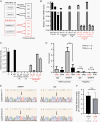
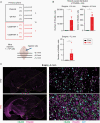
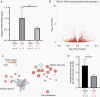
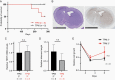
Results: Orthotopic injection of neuronal precursor cells (NPCs) derived from hiPSCs with the TPM into immunodeficient mice did not enhance tumorigenesis compared to TERT promoter wild type NPCs at initial in vivo passage presumably due to relatively long telomeres. However, the mutation recruited GA-Binding Protein and engendered low-level TERT expression resulting in enhanced tumorigenesis and maintenance of short telomeres upon secondary passage as observed in human GBM. These results provide the first insights regarding increased tumorigenesis upon introducing a TPM compared to isogenic controls without TPMs.
Conclusion: Our novel GBM models presented the growth advantage of heterozygous TPMs for the first time in the context of GBM driver mutations relative to isogenic controls, thereby allowing for the identification and validation of TERT promoter-specific vulnerabilities in a genetically accurate background.
Keywords: TERT promoter; genome editing; glioma; neural progenitor cell; telomerase.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Society for Neuro-Oncology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures

Comment in
-
A promising preclinical model for TERT promoter mutation in glioblastoma.Neuro Oncol. 2022 Dec 1;24(12):2076-2077. doi: 10.1093/neuonc/noac167. Neuro Oncol. 2022. PMID: 35780432 Free PMC article. No abstract available.
Similar articles
-
TERT promoter mutations and telomeres during tumorigenesis.Curr Opin Genet Dev. 2020 Feb;60:56-62. doi: 10.1016/j.gde.2020.02.001. Epub 2020 Mar 9. Curr Opin Genet Dev. 2020. PMID: 32163830 Review.
-
Mutations in the promoter of the telomerase gene TERT contribute to tumorigenesis by a two-step mechanism.Science. 2017 Sep 29;357(6358):1416-1420. doi: 10.1126/science.aao0535. Epub 2017 Aug 17. Science. 2017. PMID: 28818973 Free PMC article.
-
When the Ends Are Really the Beginnings: Targeting Telomerase for Treatment of GBM.Curr Neurol Neurosci Rep. 2018 Mar 10;18(4):15. doi: 10.1007/s11910-018-0825-7. Curr Neurol Neurosci Rep. 2018. PMID: 29525892 Review.
-
Prognostic quality of activating TERT promoter mutations in glioblastoma: interaction with the rs2853669 polymorphism and patient age at diagnosis.Neuro Oncol. 2015 Sep;17(9):1231-40. doi: 10.1093/neuonc/nov010. Epub 2015 Feb 13. Neuro Oncol. 2015. PMID: 25681309 Free PMC article.
-
TERT promoter mutation in adult granulosa cell tumor of the ovary.Mod Pathol. 2018 Jul;31(7):1107-1115. doi: 10.1038/s41379-018-0007-9. Epub 2018 Feb 15. Mod Pathol. 2018. PMID: 29449679
Cited by
-
Stem cell modeling of nervous system tumors.Dis Model Mech. 2024 Feb 1;17(2):dmm050533. doi: 10.1242/dmm.050533. Epub 2024 Feb 14. Dis Model Mech. 2024. PMID: 38353122 Free PMC article. Review.
-
Modeling brain and neural crest neoplasms with human pluripotent stem cells.Neuro Oncol. 2023 Jul 6;25(7):1225-1235. doi: 10.1093/neuonc/noad034. Neuro Oncol. 2023. PMID: 36757217 Free PMC article. Review.
-
GABP couples oncogene signaling to telomere regulation in TERT promoter mutant cancer.Cell Rep. 2022 Sep 20;40(12):111344. doi: 10.1016/j.celrep.2022.111344. Cell Rep. 2022. PMID: 36130485 Free PMC article.
-
Telomerase reactivation induces progression of mouse Braf V600E -driven thyroid cancers without telomere lengthening.bioRxiv [Preprint]. 2023 Jan 24:2023.01.24.525280. doi: 10.1101/2023.01.24.525280. bioRxiv. 2023. Update in: Mol Cancer Res. 2023 Nov 1;21(11):1163-1175. doi: 10.1158/1541-7786.MCR-23-0144. PMID: 36747657 Free PMC article. Updated. Preprint.
-
Advancing Glioblastoma Research with Innovative Brain Organoid-Based Models.Cells. 2025 Feb 16;14(4):292. doi: 10.3390/cells14040292. Cells. 2025. PMID: 39996764 Free PMC article. Review.
References
-
- Arita H, Narita Y, Fukushima S, et al. . Upregulating mutations in the TERT promoter commonly occur in adult malignant gliomas and are strongly associated with total 1p19q loss. Acta Neuropathol. 2013; 126(2):267–276. - PubMed
-
- Körber V, Yang J, Barah P, et al. . Evolutionary trajectories of IDHWT glioblastomas reveal a common path of early tumorigenesis instigated years ahead of initial diagnosis. Cancer Cell. 2019; 35(4):692–704.e12. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

