Thiol Reductases in Deinococcus Bacteria and Roles in Stress Tolerance
- PMID: 35326211
- PMCID: PMC8945050
- DOI: 10.3390/antiox11030561
Thiol Reductases in Deinococcus Bacteria and Roles in Stress Tolerance
Abstract
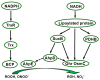
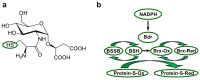
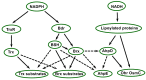
Deinococcus species possess remarkable tolerance to extreme environmental conditions that generate oxidative damage to macromolecules. Among enzymes fulfilling key functions in metabolism regulation and stress responses, thiol reductases (TRs) harbour catalytic cysteines modulating the redox status of Cys and Met in partner proteins. We present here a detailed description of Deinococcus TRs regarding gene occurrence, sequence features, and physiological functions that remain poorly characterised in this genus. Two NADPH-dependent thiol-based systems are present in Deinococcus. One involves thioredoxins, disulfide reductases providing electrons to protein partners involved notably in peroxide scavenging or in preserving protein redox status. The other is based on bacillithiol, a low-molecular-weight redox molecule, and bacilliredoxin, which together protect Cys residues against overoxidation. Deinococcus species possess various types of thiol peroxidases whose electron supply depends either on NADPH via thioredoxins or on NADH via lipoylated proteins. Recent data gained on deletion mutants confirmed the importance of TRs in Deinococcus tolerance to oxidative treatments, but additional investigations are needed to delineate the redox network in which they operate, and their precise physiological roles. The large palette of Deinococcus TR representatives very likely constitutes an asset for the maintenance of redox homeostasis in harsh stress conditions.
Keywords: Deinococcus; bacillithiol; cysteine; oxidative stress; peroxidase; protein redox status; reductase; thiol; thioredoxin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
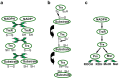
References
-
- de Groot A., Dulermo R., Ortet P., Blanchard L., Guerin P., Fernandez B., Vacherie B., Dossat C., Jolivet E., Siguier P., et al. Alliance of Proteomics and Genomics to Unravel the Specificities of Sahara Bacterium Deinococcus deserti. PLoS Genet. 2009;5:e1000434. doi: 10.1371/journal.pgen.1000434. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

