Smo-Shh Agonist Purmorphamine Prevents Neurobehavioral and Neurochemical Defects in 8-OH-DPAT-Induced Experimental Model of Obsessive-Compulsive Disorder
- PMID: 35326298
- PMCID: PMC8946713
- DOI: 10.3390/brainsci12030342
Smo-Shh Agonist Purmorphamine Prevents Neurobehavioral and Neurochemical Defects in 8-OH-DPAT-Induced Experimental Model of Obsessive-Compulsive Disorder
Abstract
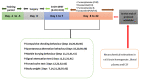
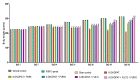
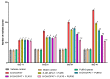
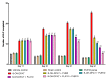
Obsessive-compulsive disorder is a mental disorder characterized by repetitive, unwanted thoughts and behavior due to abnormal neuronal corticostriatal-thalamocortical pathway and other neurochemical changes. Purmorphamine is a smoothened-sonic-hedgehog agonist that has a protective effect against many neurological diseases due to its role in maintaining functional connectivity during CNS development and its anti-inflammatory and antioxidant properties. As part of our current research, we investigated the neuroprotective effects of PUR against behavioral and neurochemical changes in 8-hydroxy-2-(di-n-propylamino)-tetralin-induced obsessive-compulsive disorder in rats. Additionally, the effect of PUR was compared with the standard drug for OCD, i.e., fluvoxamine. The intra-dorsal raphe-nucleus injection of 8-OH-DPAT in rats for seven days significantly showed OCD-like repetitive and compulsive behavior along with increased oxidative stress, inflammation, apoptosis, as well as neurotransmitter imbalance. These alterations were dose-dependently attenuated by long-term purmorphamine treatment at 5 mg/kg and 10 mg/kg i.p. In this study, we assessed the level of various neurochemical parameters in different biological samples, including brain homogenate, blood plasma, and CSF, to check the drug's effect centrally and peripherally. These effects were comparable to the standard oral treatment withfluvoxamine at 10 mg/kg. However, when fluvoxamine was given in combination with purmorphamine, there was a more significant restoration of these alterations than the individualtreatmentswithfluvoxamine and purmorphamine. All the above findings demonstrate that the neuroprotective effect of purmorphamine in OCD can be strong evidence for developing a new therapeutic target for treating and managing OCD.
Keywords: 8-OH-DPAT; Smo-Shh; fluvoxamine; obsessive-compulsive disorder; purmorphamine; serotonin.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures



Similar articles
-
Acetyl-11-keto-beta boswellic acid(AKBA) modulates CSTC-pathway by activating SIRT-1/Nrf2-HO-1 signalling in experimental rat model of obsessive-compulsive disorder: Evidenced by CSF, blood plasma and histopathological alterations.Neurotoxicology. 2023 Sep;98:61-85. doi: 10.1016/j.neuro.2023.08.001. Epub 2023 Aug 5. Neurotoxicology. 2023. PMID: 37549874
-
Smo-Shh signaling activator purmorphamine ameliorates neurobehavioral, molecular, and morphological alterations in an intracerebroventricular propionic acid-induced experimental model of autism.Hum Exp Toxicol. 2021 Nov;40(11):1880-1898. doi: 10.1177/09603271211013456. Epub 2021 Apr 28. Hum Exp Toxicol. 2021. PMID: 33906504
-
Role of Sonic Hedgehog Signaling Activation in the Prevention of Neurological Abnormalities Associated with Obsessive-Compulsive Disorder.Neurotox Res. 2022 Dec;40(6):1718-1738. doi: 10.1007/s12640-022-00586-4. Epub 2022 Oct 22. Neurotox Res. 2022. PMID: 36272053 Review.
-
Purmorphamine, a Smo-Shh/Gli Activator, Promotes Sonic Hedgehog-Mediated Neurogenesis and Restores Behavioural and Neurochemical Deficits in Experimental Model of Multiple Sclerosis.Neurochem Res. 2024 Jun;49(6):1556-1576. doi: 10.1007/s11064-023-04082-9. Epub 2023 Dec 30. Neurochem Res. 2024. PMID: 38160216
-
Animal models of obsessive-compulsive disorder: rationale to understanding psychobiology and pharmacology.Psychiatr Clin North Am. 2006 Jun;29(2):371-90. doi: 10.1016/j.psc.2006.02.007. Psychiatr Clin North Am. 2006. PMID: 16650714 Review.
Cited by
-
Beta-Boswellic Acid Reverses 3-Nitropropionic Acid-Induced Molecular, Mitochondrial, and Histopathological Defects in Experimental Rat Model of Huntington's Disease.Biomedicines. 2022 Nov 9;10(11):2866. doi: 10.3390/biomedicines10112866. Biomedicines. 2022. PMID: 36359390 Free PMC article.
-
Icariin prevents methylmercury-induced experimental neurotoxicity: Evidence from cerebrospinal fluid, blood plasma, brain samples, and in-silico investigations.Heliyon. 2024 Jan 6;10(1):e24050. doi: 10.1016/j.heliyon.2024.e24050. eCollection 2024 Jan 15. Heliyon. 2024. PMID: 38226245 Free PMC article.
-
Sonic Hedgehog signaling in spinal cord injury: mechanisms and therapeutic implications.Front Mol Neurosci. 2025 Jul 30;18:1624501. doi: 10.3389/fnmol.2025.1624501. eCollection 2025. Front Mol Neurosci. 2025. PMID: 40808911 Free PMC article. Review.
-
PI3K/AKT/mTOR signalling inhibitor chrysophanol ameliorates neurobehavioural and neurochemical defects in propionic acid-induced experimental model of autism in adult rats.Metab Brain Dis. 2022 Aug;37(6):1909-1929. doi: 10.1007/s11011-022-01026-0. Epub 2022 Jun 10. Metab Brain Dis. 2022. PMID: 35687217
-
Recent advances on small molecules in osteogenic differentiation of stem cells and the underlying signaling pathways.Stem Cell Res Ther. 2022 Nov 12;13(1):518. doi: 10.1186/s13287-022-03204-4. Stem Cell Res Ther. 2022. PMID: 36371202 Free PMC article. Review.
References
-
- Viol K., Schiepek G., Kronbichler M., Hartl A., Grafetstätter C., Strasser P., Kastinger A., Schöller H., Reiter E.M., Said-Yürekli S., et al. Multi-level assessment of obsessive-compulsive disorder (OCD) reveals relations between neural and neurochemical levels. BMC Psychiatry. 2020;20:559. doi: 10.1186/s12888-020-02913-5. - DOI - PMC - PubMed
-
- Westenberg H.G.M., Fineberg N.A., Denys D. Neurobiology of Obsessive-Compulsive Disorder: Serotonin and Beyond. CNS Spectrums. 2007;12:14–27. doi: 10.1017/S1092852900002479. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

