Thymoquinone Radiosensitizes Human Colorectal Cancer Cells in 2D and 3D Culture Models
- PMID: 35326517
- PMCID: PMC8945905
- DOI: 10.3390/cancers14061363
Thymoquinone Radiosensitizes Human Colorectal Cancer Cells in 2D and 3D Culture Models
Erratum in
-
Correction: Al Bitar et al. Thymoquinone Radiosensitizes Human Colorectal Cancer Cells in 2D and 3D Culture Models. Cancers 2022, 14, 1363.Cancers (Basel). 2025 Jul 24;17(15):2449. doi: 10.3390/cancers17152449. Cancers (Basel). 2025. PMID: 40805295 Free PMC article.
Abstract
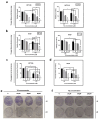
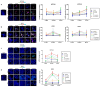
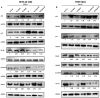
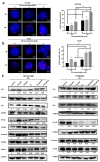
Resistance of cancer cells and normal tissue toxicity of ionizing radiation (IR) are known to limit the success of radiotherapy. There is growing interest in using IR with natural compounds to sensitize cancer cells and spare healthy tissues. Thymoquinone (TQ) was shown to radiosensitize several cancers, yet no studies have investigated its radiosensitizing effects on colorectal cancer (CRC). Here, we combined TQ with IR and determined its effects in two-dimensional (2D) and three-dimensional (3D) culture models derived from HCT116 and HT29 CRC cells, and in patient-derived organoids (PDOs). TQ sensitized CRC cells to IR and reduced cell viability and clonogenic survival and was non-toxic to non-tumorigenic intestinal cells. TQ sensitizing effects were associated with G2/M arrest and DNA damage as well as changes in key signaling molecules involved in this process. Combining a low dose of TQ (3 µM) with IR (2 Gy) inhibited sphere formation by 100% at generation 5 and this was associated with inhibition of stemness and DNA repair. These doses also led to ~1.4- to ~3.4-fold decrease in organoid forming ability of PDOs. Our findings show that combining TQ and IR could be a promising therapeutic strategy for eradicating CRC cells.
Keywords: DNA repair; cancer stem cells; colon spheres; colorectal cancer; patient-derived organoids; radiosensitization.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Silk-Ovarioids: establishment and characterization of a human ovarian primary cell 3D-model system.Hum Reprod Open. 2025 Jul 10;2025(3):hoaf042. doi: 10.1093/hropen/hoaf042. eCollection 2025. Hum Reprod Open. 2025. PMID: 40799620 Free PMC article.
-
Can a Liquid Biopsy Detect Circulating Tumor DNA With Low-passage Whole-genome Sequencing in Patients With a Sarcoma? A Pilot Evaluation.Clin Orthop Relat Res. 2025 Jan 1;483(1):39-48. doi: 10.1097/CORR.0000000000003161. Epub 2024 Jun 21. Clin Orthop Relat Res. 2025. PMID: 38905450
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Antidepressants for pain management in adults with chronic pain: a network meta-analysis.Health Technol Assess. 2024 Oct;28(62):1-155. doi: 10.3310/MKRT2948. Health Technol Assess. 2024. PMID: 39367772 Free PMC article.
Cited by
-
Potential anticancer properties and mechanisms of thymoquinone in colorectal cancer.Cancer Cell Int. 2023 Dec 12;23(1):320. doi: 10.1186/s12935-023-03174-4. Cancer Cell Int. 2023. PMID: 38087345 Free PMC article. Review.
-
Molecular Mechanisms and Signaling Pathways Underlying the Therapeutic Potential of Thymoquinone Against Colorectal Cancer.Molecules. 2024 Dec 14;29(24):5907. doi: 10.3390/molecules29245907. Molecules. 2024. PMID: 39769996 Free PMC article. Review.
-
Applications and perspectives of tumor organoids in radiobiology (Review).Oncol Rep. 2024 Aug;52(2):100. doi: 10.3892/or.2024.8759. Epub 2024 Jun 21. Oncol Rep. 2024. PMID: 38904192 Free PMC article. Review.
-
Anticancer effect of terpenes: focus on malignant melanoma.Pharmacol Rep. 2023 Oct;75(5):1115-1125. doi: 10.1007/s43440-023-00512-1. Epub 2023 Jul 29. Pharmacol Rep. 2023. PMID: 37515699 Free PMC article. Review.
-
Tumor-infiltrating immune cell signature score reveals prognostic biomarkers and therapeutic targets for colorectal cancer.Front Immunol. 2025 May 14;16:1583327. doi: 10.3389/fimmu.2025.1583327. eCollection 2025. Front Immunol. 2025. PMID: 40438100 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources

