Locoregional Treatments for Metastatic Gastrointestinal Stromal Tumor in British Columbia: A Retrospective Cohort Study from January 2008 to December 2017
- PMID: 35326632
- PMCID: PMC8945875
- DOI: 10.3390/cancers14061477
Locoregional Treatments for Metastatic Gastrointestinal Stromal Tumor in British Columbia: A Retrospective Cohort Study from January 2008 to December 2017
Abstract
Introduction: The role of surgery and non-surgical locoregional treatments (LRT) such as radiation therapy (RT) and local ablation techniques in patients with metastatic gastrointestinal stromal tumor (GIST) is unclear. This study examines LRT practice patterns in metastatic GIST and their clinical outcomes in British Columbia (BC).
Methods: Patients diagnosed with either recurrent or de novo metastatic GIST from January 2008 to December 2017 were identified. Clinical characteristics and outcomes were analyzed in patients who underwent LRT, including surgical resection of the primary tumor or metastectomy, RT, or other local ablative procedures.
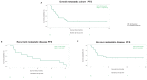
Results: 127 patients were identified: 52 (41%) had de novo metastasis and 75 (59%) had recurrent metastasis. Median age was 67 (23-90 years), 58.2% were male, primary site was 33.1% stomach, 40.2% small intestine, 11% rectum/pelvis, and 15.7% others. 37 (29.1%) of patients received palliative surgery, the majority of which had either primary tumor removal only (43.3%) or both primary tumor removal and metastectomy (35.1%). A minority of patients underwent metastectomy only (21.6%). A total of 12 (9.5%) patients received palliative RT to metastatic sites only (58.3%) or primary tumors only (41.7%), mostly for symptomatic control (n = 9). A few patients (n = 3) received local ablation for liver metastatic deposits with 1 patient receiving microwave ablation (MWA) and 2 receiving radiofrequency ablation (RFA). Most patients (n = 120, 94.5%) received some type of systemic treatment. It is notable that prolonged progression free survival (PFS) was observed for the majority of patients who underwent surgery in the metastatic setting with a median PFS of 20.5 (95% confidence interval (CI): 14.29-40.74) months. In addition, significantly higher median overall survival (mOS) was observed in patients who underwent surgery (97.15 months; 95% CI: 77.7-not reached) and LRT (78.98 months; 95% CI: 65.58-not reached) versus no surgery (45.37 months; 95% CI: 38.7-64.69) and no LRT (45.27 months; 95% CI: 33.25-58.66). Almost all patients (8 out of 9) achieved symptomatic improvement after palliative RT. All 3 patients achieved partial response and 2 out of 3 patients had relatively durable responses of 1 year or more after local ablation.
Discussion: This study is among the first to systematically examine the use of various LRT in metastatic GIST management. Integration of LRT with systemic treatments may potentially provide promising durable response and prolonged survival for highly selected metastatic GIST patients with low volume disease, limited progression and otherwise well controlled on systemic treatments. These observations, consistent with others, add to the growing evidence that supports the judicious use of LRT in combination with systemic treatments to further optimize the care of metastatic GIST patients.
Keywords: Tyrosine Kinase Inhibitor (TKI); gastrointestinal stromal tumor (GIST); local ablation; localregional treatment (LRT); radiation treatment (RT); surgery.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
[Clinicopathological features and prognosis of gastrointestinal stromal tumors with KIT/PDGFRA gene "homozygous mutation": a multicenter retrospective cohort study].Zhonghua Wei Chang Wai Ke Za Zhi. 2021 Sep 25;24(9):804-813. doi: 10.3760/cma.j.cn.441530-20210720-00293. Zhonghua Wei Chang Wai Ke Za Zhi. 2021. PMID: 34530562 Chinese.
-
Utilization of Mutational Analysis (MA) in Gastrointestinal Stromal Tumor (GIST) Management in British Columbia (BC) Between January 2008 to December 2017: a Retrospective Population-Based Study.J Gastrointest Cancer. 2022 Sep;53(3):709-717. doi: 10.1007/s12029-021-00682-2. Epub 2021 Sep 6. J Gastrointest Cancer. 2022. PMID: 34486087
-
Multimodal Treatment in Metastatic Colorectal Cancer (mCRC) Improves Outcomes-The University College London Hospital (UCLH) Experience.Cancers (Basel). 2020 Nov 27;12(12):3545. doi: 10.3390/cancers12123545. Cancers (Basel). 2020. PMID: 33261002 Free PMC article.
-
Surgical Management of Metastatic Gastrointestinal Stromal Tumors.Curr Treat Options Oncol. 2021 Mar 20;22(5):37. doi: 10.1007/s11864-021-00837-0. Curr Treat Options Oncol. 2021. PMID: 33743084 Review.
-
[Advanced gastrointestinal stromal tumors : What role does surgery currently play in multimodal concepts?].Chirurg. 2016 May;87(5):389-97. doi: 10.1007/s00104-016-0180-7. Chirurg. 2016. PMID: 27080051 Review. German.
Cited by
-
Palliative Radiation Therapy without Chemotherapy for a Patient with Monomorphic Epitheliotropic Intestinal T Cell Lymphoma: A Case Report.Palliat Med Rep. 2022 Nov 3;3(1):272-278. doi: 10.1089/pmr.2022.0011. eCollection 2022. Palliat Med Rep. 2022. PMID: 36876294 Free PMC article.
-
Evolution of Patterns of Care and Outcomes in the Real-Life Setting for Patients with Metastatic GIST Treated in Three French Expert Centers over Three Decades.Cancers (Basel). 2023 Aug 28;15(17):4306. doi: 10.3390/cancers15174306. Cancers (Basel). 2023. PMID: 37686582 Free PMC article.
-
Pan-Canadian consensus recommendations for GIST management in high- and low-throughput centres across Canada.Ther Adv Med Oncol. 2024 Aug 2;16:17588359241266179. doi: 10.1177/17588359241266179. eCollection 2024. Ther Adv Med Oncol. 2024. PMID: 39386314 Free PMC article. Review.
-
Evaluation of Systemic Treatment Options for Gastrointestinal Stromal Tumours.Cancers (Basel). 2023 Aug 13;15(16):4081. doi: 10.3390/cancers15164081. Cancers (Basel). 2023. PMID: 37627109 Free PMC article. Review.
-
Radiotherapy in the Management of Gastrointestinal Stromal Tumors: A Systematic Review.Cancers (Basel). 2022 Jun 28;14(13):3169. doi: 10.3390/cancers14133169. Cancers (Basel). 2022. PMID: 35804945 Free PMC article. Review.
References
-
- Demetri G.D., Von Mehren M., Blanke C.D., Van den Abbeele A.D., Eisenberg B., Roberts P.J., Heinrich M.C., Tuveson D.A., Singer S., Janicek M., et al. Efficacy and Safety of Imatinib Mesylate in Advanced Gastrointestinal Stromal Tumors. N. Engl. J. Med. 2002;347:472–480. doi: 10.1056/NEJMoa020461. - DOI - PubMed
-
- Joensuu H., Roberts P.J., Sarlomo-Rikala M., Andersson L.C., Tervahartiala P., Tuveson D., Silberman S.L., Capdeville R., Dimitrijevic S., Druker B., et al. Effect of the Tyrosine Kinase Inhibitor STI571 in a Patient with a Metastatic Gastrointestinal Stromal Tumor. N. Engl. J. Med. 2001;344:1052–1056. doi: 10.1056/NEJM200104053441404. - DOI - PubMed
-
- Demetri G.D., Reichardt P., Kang Y.-K., Blay J.-Y., Rutkowski P., Gelderblom H., Hohenberger P., Leahy M., Von Mehren M., Joensuu H., et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:295–302. doi: 10.1016/S0140-6736(12)61857-1. - DOI - PMC - PubMed
-
- Demetri G.D., van Oosterom A.T., Garrett C.R., Blackstein M.E., Shah M.H., Verweij J., McArthur G., Judson I.R., Heinrich M.C., Morgan J.A., et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: A randomised controlled trial. Lancet. 2006;368:1329–1338. doi: 10.1016/S0140-6736(06)69446-4. - DOI - PubMed
LinkOut - more resources
Full Text Sources

