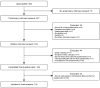
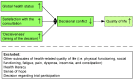
Decisional Conflict after Deciding on Potential Participation in Early Phase Clinical Cancer Trials: Dependent on Global Health Status, Satisfaction with Communication, and Timing
- PMID: 35326653
- PMCID: PMC8946532
- DOI: 10.3390/cancers14061500
Decisional Conflict after Deciding on Potential Participation in Early Phase Clinical Cancer Trials: Dependent on Global Health Status, Satisfaction with Communication, and Timing
Abstract
When standard treatment options are not available anymore, patients with advanced cancer may participate in early phase clinical trials. Improving this complex decision-making process may improve their quality of life. Therefore, this prospective multicenter study with questionnaires untangles several contributing factors to decisional conflict (which reflects the quality of decision-making) in patients with advanced cancer who recently decided upon early phase clinical trial participation (phase I or I/II). We hypothesized that health-related quality of life, health literacy, sense of hope, satisfaction with the consultation, timing of the decision, and the decision explain decisional conflict. Mean decisional conflict in 116 patients was 30.0 (SD = 16.9). Multivariate regression analysis showed that less decisional conflict was reported by patients with better global health status (β = −0.185, p = 0.018), higher satisfaction (β = −0.246, p = 0.002), and who made the decision before (β = −0.543, p < 0.001) or within a week after the consultation (β = −0.427, p < 0.001). These variables explained 37% of the variance in decisional conflict. Healthcare professionals should realize that patients with lower global health status and who need more time to decide may require additional support. Although altering such patient intrinsic characteristics is difficult, oncologists can impact the satisfaction with the consultation. Future research should verify whether effective patient-centered communication could prevent decisional conflict.
Keywords: decision making; early phase clinical trials; health literacy; hope; patient satisfaction; quality of life.
Conflict of interest statement
C.C.D.v.d.R. has received the grant from Dutch Cancer Society for the conduct of the study, and a personal fee from Kyowa Kirin BV and Ipsen Pharmaceutica BV (both outside the submitted work). M.P.L. reports grants from Astellas Pharma BV, Janssen Cilag BV, Sanofi Aventis Netherlands BV, Merck Sharp, and Dohme BV, and payment or honoraria from Amgen, Janssen Cilag, Bayer, Servier, Roche, INCa, Pfizer, Astellas, Astra Zeneca, Merck, Novartis, and Julius Clinical (all outside the submitted work). The other authors declare no conflict of interest.
Figures
Similar articles
-
Realizing better doctor-patient dialogue about choices in palliative care and early phase clinical trial participation: towards an online value clarification tool (OnVaCT).BMC Palliat Care. 2019 Nov 29;18(1):106. doi: 10.1186/s12904-019-0486-6. BMC Palliat Care. 2019. PMID: 31783851 Free PMC article.
-
Decisional Conflict During Major Medical Treatment Decision-making: a Survey Study.J Gen Intern Med. 2021 Jan;36(1):55-61. doi: 10.1007/s11606-020-06125-1. Epub 2020 Aug 17. J Gen Intern Med. 2021. PMID: 32808213 Free PMC article.
-
Factors influencing patients' decision-making about preimplantation genetic testing for monogenic disorders.Hum Reprod. 2022 Oct 31;37(11):2599-2610. doi: 10.1093/humrep/deac185. Hum Reprod. 2022. PMID: 36006036
-
Shared Decision Making in Surgery: A Meta-Analysis of Existing Literature.Patient. 2020 Dec;13(6):667-681. doi: 10.1007/s40271-020-00443-6. Patient. 2020. PMID: 32880820 Review.
-
Ethical case interventions for adult patients.Cochrane Database Syst Rev. 2019 Jul 22;7(7):CD012636. doi: 10.1002/14651858.CD012636.pub2. Cochrane Database Syst Rev. 2019. PMID: 31424106 Free PMC article. Review.
Cited by
-
Improving Shared Decision-Making in Early Phase Clinical Trials and Palliative Care: A Prospective Study on the Impact of an Online Value Clarification Tool Intervention.Psychooncology. 2025 May;34(5):e70168. doi: 10.1002/pon.70168. Psychooncology. 2025. PMID: 40302152 Free PMC article.
-
Understanding the Role of Patient-Reported Outcomes for Decision-Making in Early-Phase Dose-Finding Clinical Trials.Curr Oncol. 2025 Mar 19;32(3):176. doi: 10.3390/curroncol32030176. Curr Oncol. 2025. PMID: 40136380 Free PMC article.
References
-
- van Lent L.G.G., Jabbarian L.J., van Gurp J., Hasselaar J., Lolkema M.P., van Weert J.C.M., van der Rijt C.C.D., de Jonge M.J.A. Identifying patient values impacting the decision whether to participate in early phase clinical cancer trials: A systematic review. Cancer Treat. Rev. 2021;98:102217. doi: 10.1016/j.ctrv.2021.102217. - DOI - PubMed
-
- Sepucha K.R., Borkhoff C.M., Lally J., Levin C.A., Matlock D.D., Ng C.J., Ropka M.E., Stacey D., Joseph-Williams N., Wills C.E., et al. Establishing the effectiveness of patient decision aids: Key constructs and measurement instruments. BMC Med. Inform. Decis. Mak. 2013;13:S12. doi: 10.1186/1472-6947-13-S2-S12. - DOI - PMC - PubMed
-
- Garvelink M.M., Boland L., Klein K., Nguyen D.V., Menear M., Bekker H.L., Eden K.B., LeBlanc A., O’Connor A.M., Stacey D. Decisional conflict scale findings among patients and surrogates making health decisions: Part II of an anniversary review. Med. Decis. Mak. 2019;39:316–327. doi: 10.1177/0272989X19851346. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources