Vitamin D Metabolites in Nonmetastatic High-Risk Prostate Cancer Patients with and without Zoledronic Acid Treatment after Prostatectomy
- PMID: 35326710
- PMCID: PMC8946001
- DOI: 10.3390/cancers14061560
Vitamin D Metabolites in Nonmetastatic High-Risk Prostate Cancer Patients with and without Zoledronic Acid Treatment after Prostatectomy
Abstract
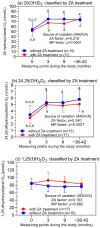
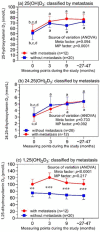
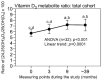
There are limited and discrepant data on prostate cancer (PCa) and vitamin D. We investigated changes in three vitamin D3 metabolites in PCa patients after prostatectomy with zoledronic acid (ZA) treatment regarding their metastasis statuses over four years. In 32 patients from the ZEUS trial, 25(OH)D3, 24,25(OH)2D3, and 1,25(OH)2D3 were measured with liquid chromatography coupled with tandem mass spectrometry at four time points. All the patients received daily calcium and vitamin D3. Bone metastases were detected in 7 of the 17 ZA-treated patients and in 5 of the 15 controls (without ZA), without differences between the groups (p = 0.725). While 25(OH)D3 and 24,25(OH)2D3 increased significantly after the study's start, with following constant values, the 1,25(OH)2D3 concentrations remained unchanged. ZA treatment did not change the levels of the three metabolites. 25(OH)D3 and 24,25(OH)2D3 were not associated with the development of bone metastases. In contrast, 1,25(OH)2D3 was also higher in patients with bone metastasis before the study's start. Thus, in high-risk PCa patients after prostatectomy, 25(OH)D3, 24,25(OH)2D3, and 1,25(OH)2D3 were not affected by supportive ZA treatment or by the development of metastasis over four years, with the exception of 1,25(OH)2D3, which was constantly higher in metastatic patients. There might be potential prognostic value if the results can be confirmed.
Keywords: 1,25(OH)2D3; 24,25(OH)2D3; 25(OH)D3; circulating vitamin D metabolites; prostate cancer; prostatectomy; vitamin D; zoledronic acid treatment.
Conflict of interest statement
F.B. and M.D. are employees of Immundiagnostik AG, Bensheim, Germany. All the authors declare that they have no conflicts of interest and no direct or indirect commercial incentives associated with publishing the manuscript.
Figures
Similar articles
-
Comparison of the effect of daily versus bolus dose maternal vitamin D3 supplementation on the 24,25-dihydroxyvitamin D3 to 25-hydroxyvitamin D3 ratio.Bone. 2018 May;110:321-325. doi: 10.1016/j.bone.2018.02.024. Epub 2018 Feb 24. Bone. 2018. PMID: 29486367 Free PMC article. Clinical Trial.
-
Association of vitamin D3 and its metabolites in patients with and without type 2 diabetes and their relationship to diabetes complications.Ther Adv Chronic Dis. 2020 Sep 26;11:2040622320924159. doi: 10.1177/2040622320924159. eCollection 2020. Ther Adv Chronic Dis. 2020. PMID: 33062234 Free PMC article.
-
1alpha(OH)D3 One-alpha-hydroxy-cholecalciferol--an active vitamin D analog. Clinical studies on prophylaxis and treatment of secondary hyperparathyroidism in uremic patients on chronic dialysis.Dan Med Bull. 2008 Nov;55(4):186-210. Dan Med Bull. 2008. PMID: 19232159 Review.
-
The effect of an acute bout of exercise on circulating vitamin D metabolite concentrations: a randomised crossover study in healthy adults.J Physiol. 2024 Sep;602(17):4157-4170. doi: 10.1113/JP286395. Epub 2024 Aug 4. J Physiol. 2024. PMID: 39097829 Clinical Trial.
-
Abiraterone inhibits 1α,25-dihydroxyvitamin D3 metabolism by CYP3A4 in human liver and intestine in vitro.J Steroid Biochem Mol Biol. 2014 Oct;144 Pt A:50-8. doi: 10.1016/j.jsbmb.2013.10.027. Epub 2013 Nov 20. J Steroid Biochem Mol Biol. 2014. PMID: 24269662 Review.
Cited by
-
Dietary Epigenetic Modulators: Unravelling the Still-Controversial Benefits of miRNAs in Nutrition and Disease.Nutrients. 2024 Jan 3;16(1):160. doi: 10.3390/nu16010160. Nutrients. 2024. PMID: 38201989 Free PMC article. Review.
References
-
- Giustina A., Adler R.A., Binkley N., Bouillon R., Ebeling P.R., Lazaretti-Castro M., Marcocci C., Rizzoli R., Sempos C.T., Bilezikian J.P. Controversies in vitamin D: Summary Statement from an International Conference. J. Clin. Endocrinol. Metab. 2019;104:234–240. doi: 10.1210/jc.2018-01414. - DOI - PubMed
-
- Giustina A., Adler R.A., Binkley N., Bollerslev J., Bouillon R., Dawson-Hughes B., Ebeling P.R., Feldman D., Formenti A.M., Lazaretti-Castro M., et al. Consensus statement from 2(nd) International Conference on Controversies in Vitamin D. Rev. Endocr. Metab. Disord. 2020;21:89–116. doi: 10.1007/s11154-019-09532-w. - DOI - PMC - PubMed
-
- Bouillon R., Marcocci C., Carmeliet G., Bikle D., White J.H., Dawson-Hughes B., Lips P., Munns C.F., Lazaretti-Castro M., Giustina A., et al. Skeletal and extraskeletal actions of vitamin D: Current evidence and outstanding questions. Endocr. Rev. 2019;40:1109–1151. doi: 10.1210/er.2018-00126. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

