Comparison of Different Self-Sampling Devices for Molecular Detection of Human Papillomavirus (HPV) and Other Sexually Transmitted Infections (STIs): A Pilot Study
- PMID: 35326937
- PMCID: PMC8950387
- DOI: 10.3390/healthcare10030459
Comparison of Different Self-Sampling Devices for Molecular Detection of Human Papillomavirus (HPV) and Other Sexually Transmitted Infections (STIs): A Pilot Study
Abstract
Background: Cervical cancer is the fourth most common cancer in women, and it is well known that high-risk human papillomavirus (hrHPV) infections are the necessary carcinogenic factors for the development of cervical tumors. Moreover, the interaction between HPV and other sexually transmitted infections (STIs) may increase the risk of cancer progression. Self-sampling has been demonstrated to represent a valid and well-accepted alternative, favoring women's participation in screening programs. This study aimed to investigate the use of FLOQSwabs® (FS) as compared to two other vaginal self-collection devices for the detection of hrHPV and other sexually transmitted infections.
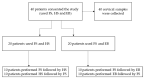
Methods: Cervical and vaginal self-samples were collected, using two different combinations of vaginal self-sampling devices, from 40 women referred to colposcopy for a documented abnormal Pap smear. All samples were tested for hrHPV and seven STI pathogens using two commercial molecular assays.
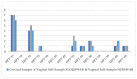
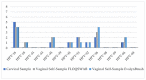
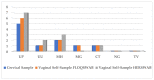
Results: Data on hrHPV detection from the first group of women showed an almost perfect agreement (kappa: 0.89) between cervical vs. FS vaginal self-samples, and a substantial agreement (kappa: 0.79) between cervical and HerSwab™ (HS) samples. In the second group of women, an almost perfect agreement (kappa: 0.90) was demonstrated in the detection of hrHPV between cervical samples vs. FS, and a moderate agreement (kappa: 0.60) for cervical vs. Evalyn®Brush (EB) self-collected samples. STI detections showed a very good agreement (kappa: 0.89 and kappa: 1.00) both among FS vs. HS and FS vs. EB, respectively. There was no statistically significant difference between the different devices used. The most frequently detected hrHPV genotypes in the studied population were HPV 16, 31, 35, 51, and 56; whilst the most frequently identified STI pathogens were Ureaplasma parvum and Mycoplasma hominis. Overall, investigated women did not report any discomfort in using the different vaginal self-collection devices.
Conclusion: Evaluation of the three different vaginal self-collection devices confirmed their overall good acceptability by the studied population, as well as a similar agreement for hrHPV detection as compared to cervical samples. Our study indicated that the use of self-collected samples offers an alternative strategy to improve women's participation in cervical cancer screening programs, but also underlined the importance of evaluating the concordance in hrHPV detection of collection devices in combination with the molecular hrHPV assay.
Keywords: HPV (human papillomavirus); acceptability; clinical accuracy; high-risk human papillomavirus (hrHPV); self-collected samples; sexually transmitted infections (STIs); vaginal self-sampling devices.
Conflict of interest statement
C.C.E. is cofounder of Hiantis Srl and has had previous research collaborations with Copan Italia, Seegene, BD, and Novosanis. S.C. is a consultant for Copan. All other authors declare no conflict of interest.
Figures
Similar articles
-
Comparison of diagnostic accuracy and acceptability of self-sampling devices for human Papillomavirus detection: A systematic review.Prev Med Rep. 2024 Jan 4;38:102590. doi: 10.1016/j.pmedr.2024.102590. eCollection 2024 Feb. Prev Med Rep. 2024. PMID: 38283967 Free PMC article. Review.
-
Molecular Detection of Human Papillomavirus (HPV) and Other Sexually Transmitted Pathogens in Cervical and Self-Collected Specimens.Int J Mol Sci. 2025 Feb 3;26(3):1296. doi: 10.3390/ijms26031296. Int J Mol Sci. 2025. PMID: 39941064 Free PMC article.
-
Prevalence of Human Papillomavirus (HPV) and Other Sexually Transmitted Infections (STIs) among Italian Women Referred for a Colposcopy.Int J Environ Res Public Health. 2019 Dec 9;16(24):5000. doi: 10.3390/ijerph16245000. Int J Environ Res Public Health. 2019. PMID: 31818033 Free PMC article.
-
Safety and acceptability of human papillomavirus testing of self-collected specimens: A methodologic study of the impact of collection devices and HPV assays on sensitivity for cervical cancer and high-grade lesions.J Clin Virol. 2018 Feb-Mar;99-100:22-30. doi: 10.1016/j.jcv.2017.12.008. Epub 2017 Dec 21. J Clin Virol. 2018. PMID: 29289814
-
Comparing Clinician-Collected and Self-Collected Tests for Detecting High-Risk HPV Infection among Female-to-Male Transgender Adults [Internet].Washington (DC): Patient-Centered Outcomes Research Institute (PCORI); 2018 Sep. Washington (DC): Patient-Centered Outcomes Research Institute (PCORI); 2018 Sep. PMID: 37844157 Free Books & Documents. Review.
Cited by
-
Sexually transmitted infections in the middle east and North Africa: comprehensive systematic review and meta-analysis.BMC Infect Dis. 2024 Nov 1;24(1):1229. doi: 10.1186/s12879-024-10153-1. BMC Infect Dis. 2024. PMID: 39487391 Free PMC article.
-
Comparison of diagnostic accuracy and acceptability of self-sampling devices for human Papillomavirus detection: A systematic review.Prev Med Rep. 2024 Jan 4;38:102590. doi: 10.1016/j.pmedr.2024.102590. eCollection 2024 Feb. Prev Med Rep. 2024. PMID: 38283967 Free PMC article. Review.
-
Innovations in healthcare delivery: Human papilloma virus self sampling diagnostics and participatory innovations for CCS.Cancer Med. 2023 Jul;12(14):15544-15551. doi: 10.1002/cam4.6201. Epub 2023 Jun 16. Cancer Med. 2023. PMID: 37325887 Free PMC article. Review.
-
Age-specific and genotype-specific carcinogenic human papillomavirus prevalence in a country with a high cervical cancer burden: results of a cross-sectional study in Estonia.BMJ Open. 2023 Jun 1;13(6):e069558. doi: 10.1136/bmjopen-2022-069558. BMJ Open. 2023. PMID: 37263686 Free PMC article.
-
Status of common sexually transmitted infection in population referred for colposcopy and correlation with human papillomavirus infection.BMC Womens Health. 2023 Nov 8;23(1):579. doi: 10.1186/s12905-023-02693-6. BMC Womens Health. 2023. PMID: 37940891 Free PMC article.
References
-
- World Health Organization GLOBOCAN 2018: Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2018. [(accessed on 23 April 2021)]. Available online: http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx.
-
- Walboomers J.M., Jacobs M.V., Manos M.M., Bosch F.X., Kummer J.A., Shah K.V., Snijders P.J., Peto J., Meijer C.J., Muñoz N. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J. Pathol. 1999;189:12–19. doi: 10.1002/(SICI)1096-9896(199909)189:1<12::AID-PATH431>3.0.CO;2-F. - DOI - PubMed
-
- Ronco G., Biggeri A., Confortini M., Naldoni C., Segnan N., Sideri M., Zappa M., Zorzi M., Calvia M., Accetta G., et al. Health technology assessment report: Ricerca del DNA di papillomavirus umano (HPV) come test primario per lo screening dei precursori del cancro del collo dell’utero [Health technology assessment report: HPV DNA based primary screening for cervical cancer precursors] Epidemiol. Prev. 2012;36((Suppl. S1)):e1-72. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

