Glucose- and Non-Glucose-Induced Mitochondrial Dysfunction in Diabetic Kidney Disease
- PMID: 35327540
- PMCID: PMC8945149
- DOI: 10.3390/biom12030351
Glucose- and Non-Glucose-Induced Mitochondrial Dysfunction in Diabetic Kidney Disease
Abstract
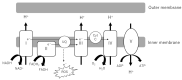
Mitochondrial dysfunction plays an important role in the pathogenesis and progression of diabetic kidney disease (DKD). In this review, we will discuss mitochondrial dysfunction observed in preclinical models of DKD as well as in clinical DKD with a focus on oxidative phosphorylation (OXPHOS), mitochondrial reactive oxygen species (mtROS), biogenesis, fission and fusion, mitophagy and urinary mitochondrial biomarkers. Both glucose- and non-glucose-induced mitochondrial dysfunction will be discussed. In terms of glucose-induced mitochondrial dysfunction, the energetic shift from OXPHOS to aerobic glycolysis, called the Warburg effect, occurs and the resulting toxic intermediates of glucose metabolism contribute to DKD-induced injury. In terms of non-glucose-induced mitochondrial dysfunction, we will review the roles of lipotoxicity, hypoxia and vasoactive pathways, including endothelin-1 (Edn1)/Edn1 receptor type A signaling pathways. Although the relative contribution of each of these pathways to DKD remains unclear, the goal of this review is to highlight the complexity of mitochondrial dysfunction in DKD and to discuss how markers of mitochondrial dysfunction could help us stratify patients at risk for DKD.
Keywords: Warburg effect; diabetic kidney disease; mitochondrial dysfunction; mitochondrial reactive oxygen species.
Conflict of interest statement
A.F. and S.M. are inventors of pending and issued patents (US10183038; US10052345; PCT/US2019/032215; PCT/US2019/041730; PCT/US2013/036484; US17/057247; US17/259883; Japan no. 501309/2021; Europe no. 19834217.2; China no. 201980060078.3; Canada no. 2852904; 2930119; 3012773) aimed at preventing and treating renal disease. They stand to gain royalties from the future commercialization of these patents. S.M. and A.F. hold equity interest in L&F Research and ZyVersa Therapeutics, Inc. which has licensed worldwide rights to develop and commercialize hydroxypropyl-beta-cyclodextrin from L&F Research for the treatment of kidney disease. A.F. also holds equities in Renal 3 River Corporation. A.F. and S.M. are supported by Aurinia Pharmaceuticals and Boehringer Ingelheim.
Figures


Comment in
-
Redox Imbalance and Mitochondrial Abnormalities in Kidney Disease.Biomolecules. 2022 Mar 21;12(3):476. doi: 10.3390/biom12030476. Biomolecules. 2022. PMID: 35327668 Free PMC article.
Similar articles
-
Glomerular Endothelial Mitochondrial Dysfunction Is Essential and Characteristic of Diabetic Kidney Disease Susceptibility.Diabetes. 2017 Mar;66(3):763-778. doi: 10.2337/db16-0695. Epub 2016 Nov 29. Diabetes. 2017. PMID: 27899487 Free PMC article.
-
Mitochondrial dysfunction in diabetic tubulopathy.Metabolism. 2022 Jun;131:155195. doi: 10.1016/j.metabol.2022.155195. Epub 2022 Mar 28. Metabolism. 2022. PMID: 35358497 Review.
-
Mitochondrial Contribution to Inflammation in Diabetic Kidney Disease.Cells. 2022 Nov 16;11(22):3635. doi: 10.3390/cells11223635. Cells. 2022. PMID: 36429063 Free PMC article. Review.
-
Broadening horizons: the contribution of mitochondria-associated endoplasmic reticulum membrane (MAM) dysfunction in diabetic kidney disease.Int J Biol Sci. 2023 Aug 21;19(14):4427-4441. doi: 10.7150/ijbs.86608. eCollection 2023. Int J Biol Sci. 2023. PMID: 37781026 Free PMC article. Review.
-
Mitochondrial dysfunction in diabetic kidney disease.Clin Chim Acta. 2019 Sep;496:108-116. doi: 10.1016/j.cca.2019.07.005. Epub 2019 Jul 2. Clin Chim Acta. 2019. PMID: 31276635 Review.
Cited by
-
Protective Factors and the Pathogenesis of Complications in Diabetes.Endocr Rev. 2024 Mar 4;45(2):227-252. doi: 10.1210/endrev/bnad030. Endocr Rev. 2024. PMID: 37638875 Free PMC article. Review.
-
Decoding diabetic kidney disease: a comprehensive review of interconnected pathways, molecular mediators, and therapeutic insights.Diabetol Metab Syndr. 2025 Jun 4;17(1):192. doi: 10.1186/s13098-025-01726-4. Diabetol Metab Syndr. 2025. PMID: 40468401 Free PMC article. Review.
-
Crosstalk of Hyperglycaemia and Cellular Mechanisms in the Pathogenesis of Diabetic Kidney Disease.Int J Mol Sci. 2024 Oct 10;25(20):10882. doi: 10.3390/ijms252010882. Int J Mol Sci. 2024. PMID: 39456664 Free PMC article. Review.
-
Role of Impaired Glycolysis in Perturbations of Amino Acid Metabolism in Diabetes Mellitus.Int J Mol Sci. 2023 Jan 15;24(2):1724. doi: 10.3390/ijms24021724. Int J Mol Sci. 2023. PMID: 36675238 Free PMC article. Review.
-
Diabetic Nephropathy: Significance of Determining Oxidative Stress and Opportunities for Antioxidant Therapies.Int J Mol Sci. 2023 Aug 3;24(15):12378. doi: 10.3390/ijms241512378. Int J Mol Sci. 2023. PMID: 37569752 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

