The Relationship between Plasma Alpha-1-Antitrypsin Polymers and Lung or Liver Function in ZZ Alpha-1-Antitrypsin-Deficient Patients
- PMID: 35327571
- PMCID: PMC8945708
- DOI: 10.3390/biom12030380
The Relationship between Plasma Alpha-1-Antitrypsin Polymers and Lung or Liver Function in ZZ Alpha-1-Antitrypsin-Deficient Patients
Abstract
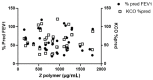
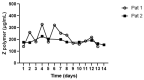
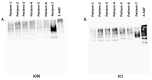
Alpha-1-Antitrypsin (AAT) is a protein of the SERPINA1 gene. A single amino acid mutation (Lys342Glu) results in an expression of misfolded Z-AAT protein, which has a high propensity to intra- and extra-cellular polymerization. Here, we asked whether levels of circulating Z-AAT polymers are associated with the severity of lung disease, liver disease, or both. We obtained cross sectional data from the Dutch part of the Alpha1 International Registry of 52 ZZ-AAT patients who performed a pulmonary function test and donated a blood sample on the same day. From the Alpha-1 Liver Aachen Registry, we obtained a cohort of 40 ZZ-AAT patients with available data on their liver function. The levels of plasma Z-AAT polymers were determined using a LG96 monoclonal antibody-based sandwich ELISA. In a Dutch cohort, the median plasma level of Z-AAT polymers of patients diagnosed for pulmonary disease was 947.5 µg/mL (733.6−1218 µg/mL (95% CI)), which did not correlate with airflow obstruction or gas transfer value. In the Alpha-1 liver patient cohort, the median polymer level was 1245.9 µg/mL (753−2034 µg/mL (95% CI)), which correlated with plasma gamma-glutamyl transferase (GGT, rs = 0.57, p = 0.001), glutamate dehydrogenase (GLDH, rs = 0.48, p = 0.002) and triglycerides (TG, rs = 0.48, p = 0.0046). A Wilcoxon rank test showed higher Z-AAT polymer values for the liver over the lung group (p < 0.0001). These correlations support a possible link between plasma Z-AAT polymers and the liver function.
Keywords: ELISA; SERPINA1; alpha1-antitrypsin; deficiency; liver biomarkers; lung function; polymers.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


Similar articles
-
Association between circulating alpha-1 antitrypsin polymers and lung and liver disease.Respir Res. 2021 Sep 15;22(1):244. doi: 10.1186/s12931-021-01842-5. Respir Res. 2021. PMID: 34526035 Free PMC article.
-
Quantification of circulating alpha-1-antitrypsin polymers associated with different SERPINA1 genotypes.Clin Chem Lab Med. 2024 Feb 27;62(10):1980-1990. doi: 10.1515/cclm-2023-1348. Print 2024 Sep 25. Clin Chem Lab Med. 2024. PMID: 38407261
-
Polymerization of misfolded Z alpha-1 antitrypsin protein lowers CX3CR1 expression in human PBMCs.Elife. 2021 May 18;10:e64881. doi: 10.7554/eLife.64881. Elife. 2021. PMID: 34002692 Free PMC article.
-
Alpha1-antitrypsin deficiency: New therapies on the horizon.Curr Opin Pharmacol. 2021 Aug;59:149-156. doi: 10.1016/j.coph.2021.06.001. Epub 2021 Jul 10. Curr Opin Pharmacol. 2021. PMID: 34256305 Review.
-
Gene therapy for alpha-1 antitrypsin deficiency.Hum Mol Genet. 2011 Apr 15;20(R1):R87-92. doi: 10.1093/hmg/ddr156. Epub 2011 Apr 16. Hum Mol Genet. 2011. PMID: 21498872 Free PMC article. Review.
Cited by
-
Capturing the conversion of the pathogenic alpha-1-antitrypsin fold by ATF6 enhanced proteostasis.Cell Chem Biol. 2023 Jan 19;30(1):22-42.e5. doi: 10.1016/j.chembiol.2022.12.004. Epub 2023 Jan 10. Cell Chem Biol. 2023. PMID: 36630963 Free PMC article.
-
Serum a-1 antitrypsin as a novel biomarker in chronic heart failure.ESC Heart Fail. 2023 Oct;10(5):2865-2874. doi: 10.1002/ehf2.14451. Epub 2023 Jul 7. ESC Heart Fail. 2023. PMID: 37417425 Free PMC article.
-
Tracing genetic diversity captures the molecular basis of misfolding disease.Nat Commun. 2024 Apr 18;15(1):3333. doi: 10.1038/s41467-024-47520-0. Nat Commun. 2024. PMID: 38637533 Free PMC article.
-
Genetic and clinical factors influencing CF-associated liver disease: the impact of SERPINA1 variants and CFTR genotypes in Romanian pediatric cystic fibrosis patients.Med Pharm Rep. 2024 Oct;97(4):429-437. doi: 10.15386/mpr-2801. Epub 2024 Oct 30. Med Pharm Rep. 2024. PMID: 39502765 Free PMC article.
-
Association of circulating Z-polymer with adverse clinical outcomes and liver fibrosis in adults with alpha-1 antitrypsin deficiency.United European Gastroenterol J. 2024 Oct;12(8):1091-1101. doi: 10.1002/ueg2.12629. Epub 2024 Jul 18. United European Gastroenterol J. 2024. PMID: 39024029 Free PMC article.
References
-
- Jonigk D., Al-Omari M., Maegel L., Muller M., Izykowski N., Hong J., Hong K., Kim S.H., Dorsch M., Mahadeva R., et al. Anti-inflammatory and immunomodulatory properties of alpha1-antitrypsin without inhibition of elastase. Proc. Natl. Acad. Sci. USA. 2013;110:15007–15012. doi: 10.1073/pnas.1309648110. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

