Cross-Talk between p53 and Wnt Signaling in Cancer
- PMID: 35327645
- PMCID: PMC8946298
- DOI: 10.3390/biom12030453
Cross-Talk between p53 and Wnt Signaling in Cancer
Abstract
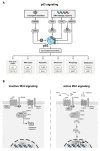
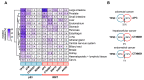
Targeting cancer hallmarks is a cardinal strategy to improve antineoplastic treatment. However, cross-talk between signaling pathways and key oncogenic processes frequently convey resistance to targeted therapies. The p53 and Wnt pathway play vital roles for the biology of many tumors, as they are critically involved in cancer onset and progression. Over recent decades, a high level of interaction between the two pathways has been revealed. Here, we provide a comprehensive overview of molecular interactions between the p53 and Wnt pathway discovered in cancer, including complex feedback loops and reciprocal transactivation. The mutational landscape of genes associated with p53 and Wnt signaling is described, including mutual exclusive and co-occurring genetic alterations. Finally, we summarize the functional consequences of this cross-talk for cancer phenotypes, such as invasiveness, metastasis or drug resistance, and discuss potential strategies to pharmacologically target the p53-Wnt interaction.
Keywords: APC; Wnt; beta-catenin; cancer; drug resistance; metastasis; p53.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures


Similar articles
-
MicroRNA-552 links Wnt signaling to p53 tumor suppressor in colorectal cancer.Int J Oncol. 2018 Oct;53(4):1800-1808. doi: 10.3892/ijo.2018.4505. Epub 2018 Jul 26. Int J Oncol. 2018. PMID: 30066856
-
Interaction of the Wnt/β-catenin and RAS-ERK pathways involving co-stabilization of both β-catenin and RAS plays important roles in the colorectal tumorigenesis.Adv Biol Regul. 2018 May;68:46-54. doi: 10.1016/j.jbior.2018.01.001. Epub 2018 Jan 10. Adv Biol Regul. 2018. PMID: 29449169 Review.
-
Missense Mutant p53 Transactivates Wnt/β-Catenin Signaling in Neighboring p53-Destabilized Cells through the COX-2/PGE2 Pathway.Cancer Res Commun. 2025 Jan 1;5(1):13-23. doi: 10.1158/2767-9764.CRC-24-0471. Cancer Res Commun. 2025. PMID: 39641656 Free PMC article.
-
p53 and microRNA-34 are suppressors of canonical Wnt signaling.Sci Signal. 2011 Nov 1;4(197):ra71. doi: 10.1126/scisignal.2001744. Sci Signal. 2011. PMID: 22045851 Free PMC article.
-
Wnt/β-catenin mediated signaling pathways in cancer: recent advances, and applications in cancer therapy.Mol Cancer. 2025 Jun 10;24(1):171. doi: 10.1186/s12943-025-02363-1. Mol Cancer. 2025. PMID: 40495229 Free PMC article. Review.
Cited by
-
Differential effect of plakoglobin in restoring the tumor suppressor activities of p53-R273H vs. p53-R175H mutants.PLoS One. 2024 Oct 3;19(10):e0306705. doi: 10.1371/journal.pone.0306705. eCollection 2024. PLoS One. 2024. PMID: 39361615 Free PMC article.
-
Pregnane X receptor activation alleviates renal fibrosis in mice via interacting with p53 and inhibiting the Wnt7a/β-catenin signaling.Acta Pharmacol Sin. 2023 Oct;44(10):2075-2090. doi: 10.1038/s41401-023-01113-7. Epub 2023 Jun 21. Acta Pharmacol Sin. 2023. PMID: 37344564 Free PMC article.
-
Overcoming Resistance to Immune Checkpoint Blockade in Liver Cancer with Combination Therapy: Stronger Together?Semin Liver Dis. 2024 May;44(2):159-179. doi: 10.1055/a-2334-8311. Epub 2024 May 28. Semin Liver Dis. 2024. PMID: 38806159 Free PMC article. Review.
-
Non-Canonical WNT/Wnt5a Pathway Activity in Circulating Monocytes of Untreated Psoriatic Patients: An Exploratory Study of Its Association with Inflammatory Cytokines and Cardiovascular Risk Marker-ADAMTS7.Biomedicines. 2023 Feb 16;11(2):577. doi: 10.3390/biomedicines11020577. Biomedicines. 2023. PMID: 36831113 Free PMC article.
-
Repurposing of US-FDA-approved drugs as negative modulators of ubiquitin specific protease-7 (USP7).Heliyon. 2024 Feb 23;10(5):e26345. doi: 10.1016/j.heliyon.2024.e26345. eCollection 2024 Mar 15. Heliyon. 2024. PMID: 38468948 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

