Calystegines Improve the Metabolic Activity of Human Adipose Derived Stromal Stem Cells (ASCs) under Hyperglycaemic Condition through the Reduction of Oxidative/ER Stress, Inflammation, and the Promotion of the AKT/PI3K/mTOR Pathway
- PMID: 35327652
- PMCID: PMC8946193
- DOI: 10.3390/biom12030460
Calystegines Improve the Metabolic Activity of Human Adipose Derived Stromal Stem Cells (ASCs) under Hyperglycaemic Condition through the Reduction of Oxidative/ER Stress, Inflammation, and the Promotion of the AKT/PI3K/mTOR Pathway
Abstract
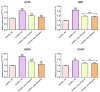
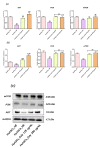
Hyperglycaemia and its resulting glucotoxicity are among the most prominent hallmarks of diabetes mellitus (DM) development. Persistent hyperglycaemia further leads to oxidative stress via mitochondrial dysfunction and subsequent ER stress onset, while associated hyperlipidaemia triggers the adipose tissue to secrete pro-inflammatory cytokines. In this study, the effect of calystegines has been investigated in an experimental model of hyperglycaemia induced on human ASCs cells. Different cellular pathways including apoptosis, oxidative and ER stress, inflammation as well as Pi3K/AKT/mTOR metabolic-associated axis have been evaluated by means on RT-qPCR, western blot, and flow cytometry techniques. Treatment of HuASCs cells with calystegines strongly promoted the hyperglycaemic cells survival and significantly diminished oxidative stress, mitochondrial dynamics failure and ER stress, while improving the endogenous cellular antioxidant defenses. Interestingly, nortropane alkaloids efficiently prevented the hyperglycaemia-mediated inflammatory response, as evidenced by the regulation of the pro- and anti-inflammatory response in HuASCs cells. Finally, we evidenced that calystegines may exert their protective effect on HuASCs cells metabolic functions through the restoration of the defective PI3K/AKT/mTOR pathway. Overall, the present investigation demonstrated that calystegines possess important abilities to protect HuASCs against hyperglycaemia-induced cellular dysfunction, and it evidenced that the observed effects are associated to the promotion of PI3K/AKT/mTOR pathway.
Keywords: ER stress; HuASCs; calystegines; hyperglycaemia; inflammation; mTOR.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

