Decreased Intrathecal Concentrations of Free Light Chains Kappa in Multiple Sclerosis Patients Taking Very High Effective Disease-Modifying Treatment
- PMID: 35328273
- PMCID: PMC8947149
- DOI: 10.3390/diagnostics12030720
Decreased Intrathecal Concentrations of Free Light Chains Kappa in Multiple Sclerosis Patients Taking Very High Effective Disease-Modifying Treatment
Abstract
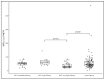
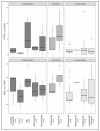
Free light chains kappa (FLCκ) in cerebrospinal fluid (CSF) are a part of the intrathecal immune response. This observational study was conducted to investigate the effects of different disease-modifying therapies (DMT) on the humoral intrathecal immune response in the CSF of patients with multiple sclerosis (MS). FLCκ were analyzed in CSF and serum samples from MS patients taking DMT (n = 60) and those in a control cohort of treatment-naïve MS patients (n = 90). DMT was classified as moderately effective (including INFß-1a, INFß-1b, glatiramer acetate, dimethyl fumarate, teriflunomide, triamcinolone); highly effective (including fingolimod, daclizumab) and very highly effective (alemtuzumab, natalizumab, rituximab/ocrelizumab, mitoxantrone). FLCκ were measured using a nephelometric FLCκ kit. Intrathecal FLCκ and IgG concentrations were assessed in relation to the hyperbolic reference range in quotient diagrams. Intrathecal FLCκ concentrations and IgG concentrations were significantly lower in samples from the cohort of MS patients taking very highly effective DMT than in samples from the cohort of MS patients taking highly effective DMT and in the treatment-naïve cohort (FLCκ: p = 0.004, p < 0.0001 respectively/IgG: p = 0.013; p = 0.021). The reduction in FLCκ could contribute to an anti-inflammatory effect in the CNS through this mechanism. There was no difference in the appearance of CSF-specific oligoclonal bands (p = 0.830). Longitudinal analyses are required to confirm these results.
Keywords: cerebrospinal fluid; disease-modifying therapy; free light chain kappa; immunoglobulin synthesis; multiple sclerosis; oligoclonal IgG.
Conflict of interest statement
The authors declare that there is no conflict of interest. Outside the submitted work, the authors received honoraria for lectures, travel grants, or research grants. M.S. reports personal fees and grants from Merck Healthcare Deutschland and Bayer Vital GmbH. M.G. received honoraria and travel reimbursements for attending meetings by Biogen, Celgene, Merck Serono, Novartis, Roche, Sanofi Genzyme, and TEVA. His research is funded by the German Ministry for Education and Research (BMBF), Merck Serono, and Novartis. TS received grants from Bristol Myers Squibb and Sanofi Aventis and personal fees from Alexion, Alnylam, Bayer Vital, Biogen, Celgene, CSL Behring, EUROIMMUN, Merck, Novartis, Roche, Sanofi Aventis, and Siemens. MN reports nonfinancial support by Siemens Healthineers, the Binding Site Group, Becton Dickinson, DZHK (German Centre for Cardiovascular Research, Partner Site Greifswald, University Medicine, Greifswald, Germany), DGKL (German Federation of Clinical Chemistry and Laboratory Medicine), German Federal Medical Association, Roche Diagnostics Germany GmbH; he also received personal fees by Boehringer Ingelheim and Becton Dickinson; Grant by DZHK, LVL Technologies, Bruker BioSpin, Abbott, Radiometer, Tosoh and IDS Immunodiagnostic Systems, Deutschland GmbH.
Figures

Similar articles
-
A New Laboratory Workflow Integrating the Free Light Chains Kappa Quotient into Routine CSF Analysis.Biomolecules. 2022 Nov 15;12(11):1690. doi: 10.3390/biom12111690. Biomolecules. 2022. PMID: 36421703 Free PMC article.
-
Implications of monoclonal gammopathy and isoelectric focusing pattern 5 on the free light chain kappa diagnostics in cerebrospinal fluid.Clin Chem Lab Med. 2024 Jul 23;63(1):147-153. doi: 10.1515/cclm-2023-1468. Print 2025 Jan 29. Clin Chem Lab Med. 2024. PMID: 39039726
-
Intrathecal B cell-related markers for an optimized biological investigation of multiple sclerosis patients.Sci Rep. 2022 Sep 30;12(1):16425. doi: 10.1038/s41598-022-19811-3. Sci Rep. 2022. PMID: 36180495 Free PMC article.
-
Multiple sclerosis: current and emerging disease-modifying therapies and treatment strategies.Mayo Clin Proc. 2014 Feb;89(2):225-40. doi: 10.1016/j.mayocp.2013.11.002. Mayo Clin Proc. 2014. PMID: 24485135 Review.
-
New FDA-Approved Disease-Modifying Therapies for Multiple Sclerosis.Clin Ther. 2015 Apr 1;37(4):691-715. doi: 10.1016/j.clinthera.2015.03.001. Epub 2015 Apr 4. Clin Ther. 2015. PMID: 25846320 Review.
Cited by
-
Kappa Free Light Chain Index Correlates With Prognostic Biomarkers in Multiple Sclerosis and Decreases Slowly Following Treatment.Eur J Neurol. 2025 Jul;32(7):e70291. doi: 10.1111/ene.70291. Eur J Neurol. 2025. PMID: 40643211 Free PMC article.
-
Kappa free light chain concentration in serum is reduced after CD20-depletion with ocrelizumab.Neurol Res Pract. 2025 Aug 22;7(1):58. doi: 10.1186/s42466-025-00419-7. Neurol Res Pract. 2025. PMID: 40847377 Free PMC article.
-
Chimeric antigen receptor T-cell therapy for autoimmune diseases of the central nervous system: a systematic literature review.J Neurol. 2024 Oct;271(10):6526-6542. doi: 10.1007/s00415-024-12642-4. Epub 2024 Sep 14. J Neurol. 2024. PMID: 39276207 Free PMC article.
-
Establishing the best combination of the kappa free light chain index and oligoclonal bands for an accurate diagnosis of multiple sclerosis.Front Immunol. 2023 Oct 25;14:1288169. doi: 10.3389/fimmu.2023.1288169. eCollection 2023. Front Immunol. 2023. PMID: 37954589 Free PMC article.
-
IgG and kappa free light chain CSF/serum indices: evaluating intrathecal immunoglobulin production in HIV infection in comparison with multiple sclerosis.Clin Chem Lab Med. 2024 Apr 4;62(10):2024-2029. doi: 10.1515/cclm-2023-1018. Print 2024 Sep 25. Clin Chem Lab Med. 2024. PMID: 38564810
References
-
- Süße M., Reiber H., Grothe M., Petersmann A., Nauck M., Dressel A., Hannich M.J. Free light chain kappa and the polyspecific immune response in MS and CIS—Application of the hyperbolic reference range for most reliable data interpretation. J. Neuroimmunol. 2020;346:577287. doi: 10.1016/j.jneuroim.2020.577287. - DOI - PubMed
-
- Schlüter M., Oswald E., Winklmeier S., Meinl I., Havla J., Eichhorn P., Meinl E., Kümpfel T. Effects of Natalizumab Therapy on Intrathecal Immunoglobulin G Production Indicate Targeting of Plasmablasts. Neurol.-Neuroimmunol. Neuroinflamm. 2021;8:e1030. doi: 10.1212/NXI.0000000000001030. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

