Simple Zn-Mediated Seleno- and Thio-Functionalization of Steroids at C-1 Position
- PMID: 35328446
- PMCID: PMC8952209
- DOI: 10.3390/ijms23063022
Simple Zn-Mediated Seleno- and Thio-Functionalization of Steroids at C-1 Position
Abstract
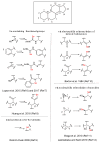
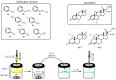
Here we report the reaction in the biphasic system of the in situ prepared selenols and thiols with 1,4-androstadiene-3,17-dione (1) or prednisone acetate (2) having α,β-unsaturated ketone as an electrophilic functionalization. The Michael-type addition reaction resulted to be chemo- and stereoselective, affording a series of novel steroidal selenides and sulfides. This is an example of a one-step, eco-friendly process that bypasses some of the main concerns connected with the bad smell and the toxicity of these seleno- and thio-reagents. Furthermore, we demonstrated that the proposed methodology offers the possibility to prepare libraries of steroids variously and selectively decorated with different organochalcogen moieties at the C1 position starting from 1,4-androstadienic skeletons and leaving unaltered the C4-C5 unsaturation. Based on the data reported in the literature the introduction of an organoselenium or an organosulfur moiety in a steroid could provide new interesting pharmaceutically active entities exerting anticancer and antimicrobial activities. In this optic, new synthetic strategies to efficiently prepare this class of compounds could be strongly desirable.
Keywords: Michael additions; selenium; steroids; sulfur; zinc.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Pacuła A.J., Mangiavacchi F., Sancineto L.J., Lenardão E., Ścianowski J., Santi C. An Update on “Selenium Containing Compounds from Poison to Drug Candidates: A Review on the GPx-like Activity”. Curr. Chem. Biol. 2016;9:97–112. doi: 10.2174/2212796810666160120220725. - DOI
-
- Mangiavacchi F., Botwina P., Menichetti E., Bagnoli L., Rosati O., Marini F., Fonseca S.F., Abenante L., Alves D., Dabrowska A., et al. Seleno-Functionalization of Quercetin Improves the Non-Covalent Inhibition of Mpro and Its Antiviral Activity in Cells against SARS-CoV-2. IJMS. 2021;22:7048. doi: 10.3390/ijms22137048. - DOI - PMC - PubMed
-
- Takafuta T., Fujimura K. Autoimmune Thrombocytopenia. Springer; Singapore: 2017. Steroids.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

