Tailoring ZE21B Alloy with Nature-Inspired Extracellular Matrix Secreted by Micro-Patterned Smooth Muscle Cells and Endothelial Cells to Promote Surface Biocompatibility
- PMID: 35328601
- PMCID: PMC8950948
- DOI: 10.3390/ijms23063180
Tailoring ZE21B Alloy with Nature-Inspired Extracellular Matrix Secreted by Micro-Patterned Smooth Muscle Cells and Endothelial Cells to Promote Surface Biocompatibility
Abstract
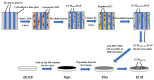
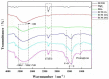
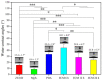
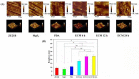
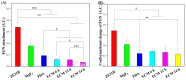
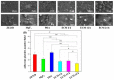
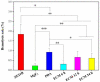
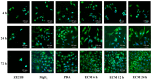
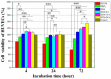
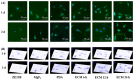
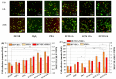
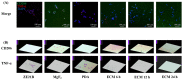
Delayed surface endothelialization is a bottleneck that restricts the further application of cardiovascular stents. It has been reported that the nature-inspired extracellular matrix (ECM) secreted by the hyaluronic acid (HA) micro-patterned smooth muscle cells (SMC) and endothelial cells (EC) can significantly promote surface endothelialization. However, this ECM coating obtained by decellularized method (dECM) is difficult to obtain directly on the surface of degradable magnesium (Mg) alloy. In this study, the method of obtaining bionic dECM by micro-patterning SMC/EC was further improved, and the nature-inspired ECM was prepared onto the Mg-Zn-Y-Nd (ZE21B) alloy surface by self-assembly. The results showed that the ECM coating not only improved surface endothelialization of ZE21B alloy, but also presented better blood compatibility, anti-hyperplasia, and anti-inflammation functions. The innovation and significance of the study is to overcome the disadvantage of traditional dECM coating and further expand the application of dECM coating to the surface of degradable materials and materials with different shapes.
Keywords: ZE21B alloy; biocompatibility; cardiovascular materials; dECM coating; hyaluronic acid micro-patterns.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- Busch R., Strohbach A., Rethfeldt S., Walz S., Busch M., Petersen S., Felix S., Sternberg K. New stent surface materials: The impact of polymer-dependent interactions of human endothelial cells, smooth muscle cells, and platelets. Acta Biomater. 2014;10:688–700. doi: 10.1016/j.actbio.2013.10.015. - DOI - PubMed
-
- Bedair T.M., Min I.J., Park W., Park B.J., Joung Y.K., Han D.K. Covalent immobilization of fibroblast-derived matrix on metallic stent for expeditious re-endothelialization. J. Ind. Eng. Chem. 2019;70:385–393. doi: 10.1016/j.jiec.2018.11.001. - DOI
-
- McFadden E.P., Stabile E., Regar E., Cheneau E., Ong A.T.L., Kinnaird T., Suddath W.O., Weissman N.J., Torguson R., Kent K.M., et al. Late thrombosis in drug-eluting coronary stents after discontinuation of antiplatelet therapy. Lancet. 2004;364:1519–1521. doi: 10.1016/S0140-6736(04)17275-9. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

