Improvements, Variations and Biomedical Applications of the Michaelis-Arbuzov Reaction
- PMID: 35328816
- PMCID: PMC8955222
- DOI: 10.3390/ijms23063395
Improvements, Variations and Biomedical Applications of the Michaelis-Arbuzov Reaction
Abstract
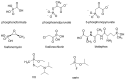
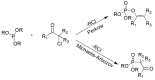
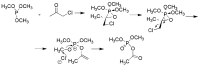
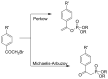
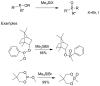
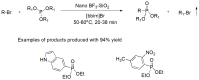
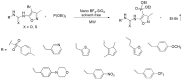
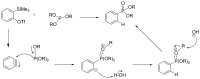
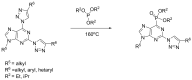
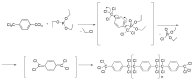
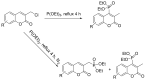
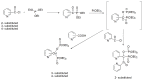
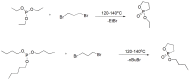
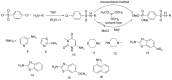
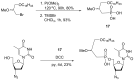
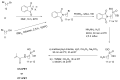
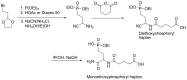
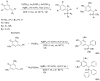
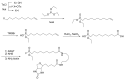
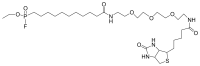
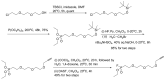
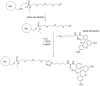
Compounds bearing the phosphorus-carbon (P-C) bond have important pharmacological, biochemical, and toxicological properties. Historically, the most notable reaction for the formation of the P-C bond is the Michaelis-Arbuzov reaction, first described in 1898. The classical Michaelis-Arbuzov reaction entails a reaction between an alkyl halide and a trialkyl phosphite to yield a dialkylalkylphosphonate. Nonetheless, deviations from the classical mechanisms and new modifications have appeared that allowed the expansion of the library of reactants and consequently the chemical space of the yielded products. These involve the use of Lewis acid catalysts, green methods, ultrasound, microwave, photochemically-assisted reactions, aryne-based reactions, etc. Here, a detailed presentation of the Michaelis-Arbuzov reaction and its developments and applications in the synthesis of biomedically important agents is provided. Certain examples of such applications include the development of alkylphosphonofluoridates as serine hydrolase inhibitors and activity-based probes, and the P-C containing antiviral and anticancer agents.
Keywords: Michaelis–Arbuzov reaction; activity-based probes; pharmaceutical applications; phosphonates; phosphonofluoridates.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
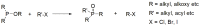
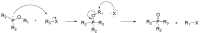
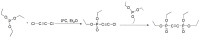
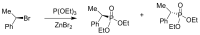
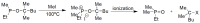
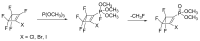
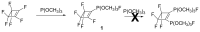
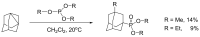
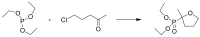
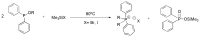
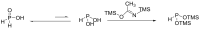
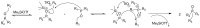
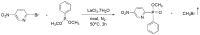
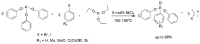

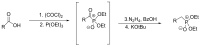

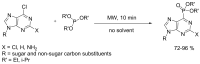
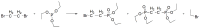
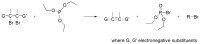




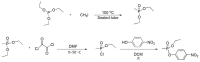
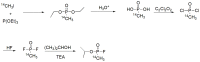
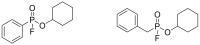

Similar articles
-
Lewis acid-mediated Michaelis-Arbuzov reaction at room temperature: a facile preparation of arylmethyl/heteroarylmethyl phosphonates.Org Lett. 2011 Mar 18;13(6):1270-3. doi: 10.1021/ol1029436. Epub 2011 Feb 24. Org Lett. 2011. PMID: 21348474
-
Cytotoxic Activity of Bisphosphonic Derivatives Obtained by the Michaelis-Arbuzov or the Pudovik Reaction.Pharmaceuticals (Basel). 2025 Jan 13;18(1):91. doi: 10.3390/ph18010091. Pharmaceuticals (Basel). 2025. PMID: 39861154 Free PMC article.
-
Solvent-Free Michaelis-Arbuzov Rearrangement under Flow Conditions.J Org Chem. 2019 Mar 1;84(5):2619-2625. doi: 10.1021/acs.joc.8b03053. Epub 2019 Feb 11. J Org Chem. 2019. PMID: 30698971
-
Recent advances in the synthesis of 4'-truncated nucleoside phosphonic acid analogues.Carbohydr Res. 2022 Mar;513:108517. doi: 10.1016/j.carres.2022.108517. Epub 2022 Feb 7. Carbohydr Res. 2022. PMID: 35152128 Review.
-
Recent Advances in the Application of P(III)-Nucleophiles to Create New P-C Bonds through Michaelis-Arbuzov-Type Rearrangement.Top Curr Chem (Cham). 2024 Mar 8;382(1):10. doi: 10.1007/s41061-024-00456-x. Top Curr Chem (Cham). 2024. PMID: 38457062 Review.
Cited by
-
Convergent Deboronative and Decarboxylative Phosphonylation Enabled by the Phosphite Radical Trap "BecaP".J Am Chem Soc. 2023 Aug 23;145(33):18649-18657. doi: 10.1021/jacs.3c06524. Epub 2023 Aug 8. J Am Chem Soc. 2023. PMID: 37552886 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

