Improved Binding Affinity of Omicron's Spike Protein for the Human Angiotensin-Converting Enzyme 2 Receptor Is the Key behind Its Increased Virulence
- PMID: 35328828
- PMCID: PMC8955673
- DOI: 10.3390/ijms23063409
Improved Binding Affinity of Omicron's Spike Protein for the Human Angiotensin-Converting Enzyme 2 Receptor Is the Key behind Its Increased Virulence
Abstract
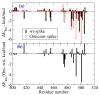
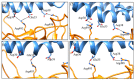
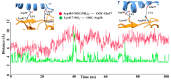
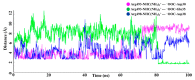
The new variant of severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2), Omicron, has been quickly spreading in many countries worldwide. Compared to the original virus, Omicron is characterized by several mutations in its genomic region, including the spike protein's receptor-binding domain (RBD). We have computationally investigated the interaction between the RBD of both the wild type and Omicron variant of SARS-CoV-2 with the human angiotensin-converting enzyme 2 (hACE2) receptor using molecular dynamics and molecular mechanics-generalized Born surface area (MM-GBSA)-based binding free energy calculations. The mode of the interaction between Omicron's RBD with the hACE2 receptor is similar to the original SARS-CoV-2 RBD except for a few key differences. The binding free energy difference shows that the spike protein of Omicron has an increased affinity for the hACE2 receptor. The mutated residues in the RBD showed strong interactions with a few amino acid residues of hACE2. More specifically, strong electrostatic interactions (salt bridges) and hydrogen bonding were observed between R493 and R498 residues of the Omicron RBD with D30/E35 and D38 residues of the hACE2, respectively. Other mutated amino acids in the Omicron RBD, e.g., S496 and H505, also exhibited hydrogen bonding with the hACE2 receptor. A pi-stacking interaction was also observed between tyrosine residues (RBD-Tyr501: hACE2-Tyr41) in the complex, which contributes majorly to the binding free energies and suggests that this is one of the key interactions stabilizing the formation of the complex. The resulting structural insights into the RBD:hACE2 complex, the binding mode information within it, and residue-wise contributions to the free energy provide insight into the increased transmissibility of Omicron and pave the way to design and optimize novel antiviral agents.
Keywords: Omicron; human angiotensin-converting enzyme 2 (hACE2); molecular dynamics simulation; molecular mechanics-generalized Born surface area (MM-GBSA); receptor-binding domain (RBD); receptor-binding motif (RBM); severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2).
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- European Centre for Disease Prevention and Control. SARS-CoV-2 Variants of Concern as of 13 January 2022. [(accessed on 10 December 2021)]. Available online: https://www.ecdc.europa.eu/en/covid-19/variants-concern.
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

