Real-World Impact of a Pharmacogenomics-Enriched Comprehensive Medication Management Program
- PMID: 35330421
- PMCID: PMC8949247
- DOI: 10.3390/jpm12030421
Real-World Impact of a Pharmacogenomics-Enriched Comprehensive Medication Management Program
Abstract
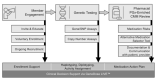
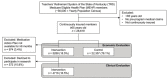
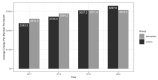
The availability of clinical decision support systems (CDSS) and other methods for personalizing medicine now allows evaluation of their real-world impact on healthcare delivery. For example, addressing issues associated with polypharmacy in older patients using pharmacogenomics (PGx) and comprehensive medication management (CMM) is thought to hold great promise for meaningful improvements across the goals of the Quadruple Aim. However, few studies testing these tools at scale, using relevant system-wide metrics, and under real-world conditions, have been published to date. Here, we document a reduction of ~$7000 per patient in direct medical charges (a total of $37 million over 5288 enrollees compared to 22,357 non-enrolled) in Medicare Advantage patients (≥65 years) receiving benefits through a state retirement system over the first 32 months of a voluntary PGx-enriched CMM program. We also observe a positive shift in healthcare resource utilization (HRU) away from acute care services and toward more sustainable and cost-effective primary care options. Together with improvements in medication risk assessment, patient/provider communication via pharmacist-mediated medication action plans (MAP), and the sustained positive trends in HRU, we suggest these results validate the use of a CDSS to unify PGx and CMM to optimize care for this and similar patient populations.
Keywords: adverse drug reactions; clinical decision support systems; comprehensive medication management; cost analysis; health resources; personalized medicine; pharmacogenomics; polypharmacy; population health; treatment outcome.
Conflict of interest statement
J.P.J., A.P.P., M.K., S.K. and J.A.S. are employees of Coriell Life Sciences and G.M.B. and Z.T.W. are employees of the Know Your Rx Coalition. V.B. was employed as a consultant by Coriell Life Sciences at the time the analysis was performed.
Figures


References
-
- U.S. Food and Drug Administration (FDA) Table of Pharmacogenomic Biomarkers in Drug Labeling. [(accessed on 11 November 2021)]; Available online: https://www.fda.gov/drugs/science-and-research-drugs/table-pharmacogenom....
LinkOut - more resources
Full Text Sources

