Synthesis of Silver Nanoparticles from Extracts of Wild Ginger (Zingiber zerumbet) with Antibacterial Activity against Selective Multidrug Resistant Oral Bacteria
- PMID: 35335369
- PMCID: PMC8949094
- DOI: 10.3390/molecules27062007
Synthesis of Silver Nanoparticles from Extracts of Wild Ginger (Zingiber zerumbet) with Antibacterial Activity against Selective Multidrug Resistant Oral Bacteria
Abstract
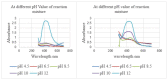
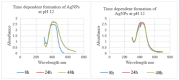
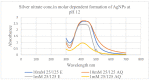
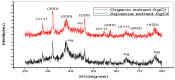
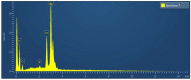
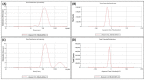
Antibiotic resistance rate is rising worldwide. Silver nanoparticles (AgNPs) are potent for fighting antimicrobial resistance (AMR), independently or synergistically. The purpose of this study was to prepare AgNPs using wild ginger extracts and to evaluate the antibacterial efficacy of these AgNPs against multidrug-resistant (MDR) Staphylococcus aureus, Streptococcus mutans, and Enterococcus faecalis. AgNPs were synthesized using wild ginger extracts at room temperature through different parameters for optimization, i.e., pH and variable molar concentration. Synthesis of AgNPs was confirmed by UV/visible spectroscopy and further characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD), energy-dispersive X-ray spectroscopy analysis (EDXA), and Fourier-transform infrared spectroscopy (FTIR). Disc and agar well diffusion techniques were utilized to determine the in vitro antibacterial activity of plant extracts and AgNPs. The surface plasmon resonance peaks in absorption spectra for silver suspension showed the absorption maxima in the range of 400-420 nm. Functional biomolecules such as N-H, C-H, O-H, C-O, and C-O-C were present in Zingiber zerumbet (Z. zerumbet) (aqueous and organic extracts) responsible for the AgNP formation characterized by FTIR. The crystalline structure of ZZAE-AgCl-NPs and ZZEE-AgCl-NPs was displayed in the XRD analysis. SEM analysis revealed the surface morphology. The EDXA analysis also confirmed the element of silver. It was revealed that AgNPs were seemingly spherical in morphology. The biosynthesized AgNPs exhibited complete antibacterial activity against the tested MDR bacterial strains. This study indicates that AgNPs of wild ginger extracts exhibit potent antibacterial activity against MDR bacterial strains.
Keywords: AgNPs; MDR pathogens; antibacterial activity; biosynthesized silver nanoparticles; wild ginger extract.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Green Synthesis and Characterization of Silver Nanoparticles Using Zingiber officinale Extracts to Investigate Their Antibacterial Potential.Int J Nanomedicine. 2024 Dec 11;19:13319-13338. doi: 10.2147/IJN.S475656. eCollection 2024. Int J Nanomedicine. 2024. PMID: 39679248 Free PMC article.
-
Characterization, Antibacterial and Antioxidant Properties of Silver Nanoparticles Synthesized from Aqueous Extracts of Allium sativum, Zingiber officinale, and Capsicum frutescens.Pharmacogn Mag. 2017 Jul;13(Suppl 2):S201-S208. doi: 10.4103/pm.pm_430_16. Epub 2017 Jul 11. Pharmacogn Mag. 2017. PMID: 28808381 Free PMC article.
-
Tuber extract of Arisaema flavum eco-benignly and effectively synthesize silver nanoparticles: Photocatalytic and antibacterial response against multidrug resistant engineered E. coli QH4.J Photochem Photobiol B. 2019 Apr;193:31-38. doi: 10.1016/j.jphotobiol.2019.01.018. Epub 2019 Feb 13. J Photochem Photobiol B. 2019. PMID: 30802773
-
Effect of Size, Shape and Surface Functionalization on the Antibacterial Activity of Silver Nanoparticles.J Funct Biomater. 2023 Apr 26;14(5):244. doi: 10.3390/jfb14050244. J Funct Biomater. 2023. PMID: 37233354 Free PMC article. Review.
-
Mechanistic aspects of plant-based silver nanoparticles against multi-drug resistant bacteria.Heliyon. 2021 Jul 1;7(7):e07448. doi: 10.1016/j.heliyon.2021.e07448. eCollection 2021 Jul. Heliyon. 2021. PMID: 34286126 Free PMC article. Review.
Cited by
-
Mechanisms of action of Sappan lignum for prostate cancer treatment: network pharmacology, molecular docking and experimental validation.Front Pharmacol. 2024 Sep 10;15:1407525. doi: 10.3389/fphar.2024.1407525. eCollection 2024. Front Pharmacol. 2024. PMID: 39318781 Free PMC article.
-
Antibiotic prescription after tooth extraction in adults: a retrospective cohort study in Austria.BMC Oral Health. 2022 Nov 20;22(1):519. doi: 10.1186/s12903-022-02556-w. BMC Oral Health. 2022. PMID: 36404324 Free PMC article.
-
Advances in silver nanoparticles: a comprehensive review on their potential as antimicrobial agents and their mechanisms of action elucidated by proteomics.Front Microbiol. 2024 Jul 31;15:1440065. doi: 10.3389/fmicb.2024.1440065. eCollection 2024. Front Microbiol. 2024. PMID: 39149204 Free PMC article. Review.
-
Green Synthesis and Characterization of Ginger-Derived Silver Nanoparticles and Evaluation of Their Antioxidant, Antibacterial, and Anticancer Activities.Plants (Basel). 2024 Apr 30;13(9):1255. doi: 10.3390/plants13091255. Plants (Basel). 2024. PMID: 38732470 Free PMC article.
-
Extracts from the Leaf of Couroupita guianensis (Aubl.): Phytochemical, Toxicological Analysis and Evaluation of Antioxidant and Antimicrobial Activities against Oral Microorganisms.Plants (Basel). 2023 Jun 15;12(12):2327. doi: 10.3390/plants12122327. Plants (Basel). 2023. PMID: 37375952 Free PMC article.
References
-
- Bae C.H., Nam S.H., Park S.M. Formation of silver nanoparticles by laser ablation of a silver target in NaCl solution. Appl. Surf. Sci. 2002;197–198:628–634. doi: 10.1016/S0169-4332(02)00430-0. - DOI
-
- Callegari A., Tonti D., Chergui M. Photochemically grown silver nanoparticles with wavelength-controlled size and shape. Nano Lett. 2003;3:1565–1568. doi: 10.1021/nl034757a. - DOI
-
- Yin B., Ma H., Wang S., Chen S. Electrochemical synthesis of silver nanoparticles under protection of poly(N-vinylpyrrolidone) J. Phys. Chem. B. 2003;107:8898–8904. doi: 10.1021/jp0349031. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

