Challenges and Complications of Poly(lactic- co-glycolic acid)-Based Long-Acting Drug Product Development
- PMID: 35335988
- PMCID: PMC8955085
- DOI: 10.3390/pharmaceutics14030614
Challenges and Complications of Poly(lactic- co-glycolic acid)-Based Long-Acting Drug Product Development
Abstract
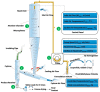
Poly(lactic-co-glycolic acid) (PLGA) is one of the preferred polymeric inactive ingredients for long-acting parenteral drug products that are constituted of complex formulations. Despite over 30 years of use, there are still many challenges faced by researchers in formulation-related aspects pertaining to drug loading and release. Until now, PLGA-based complex generic drug products have not been successfully developed. The complexity in developing these generic drug products is not just due to their complex formulation, but also to the manufacturing process of the listed reference drugs that involve PLGA. The composition and product attributes of commercial PLGA formulations vary with the drugs and their intended applications. The lack of standard compendial methods for in vitro release studies hinders generic pharmaceutical companies in their efforts to develop PLGA-based complex generic drug products. In this review, we discuss the challenges faced in developing PLGA-based long-acting injectable/implantable (LAI) drug products; hurdles that are associated with drug loading and release that are dictated by the physicochemical properties of PLGA and product manufacturing processes. Approaches to overcome these challenges and hurdles are highlighted specifically with respect to drug encapsulation and release.
Keywords: PLGA microspheres; complex generic drug products; drug delivery; long acting injectable/implantable; poly(lactide-co-glycolide); sustained release.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
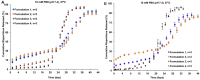
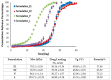
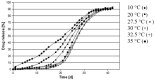
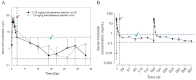



References
-
- Wang Y., Qu W., Choi S.H. FDA’s Regulatory Science Program for Generic PLA/PLGA-Based Drug Products|American Pharmaceutical Review—The Review of American Pharmaceutical Business & Technology. [(accessed on 22 October 2020)]. Available online: https://www.americanpharmaceuticalreview.com/Featured-Articles/188841-FD...
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

