Advanced Diffusion MRI of the Visual System in Glaucoma: From Experimental Animal Models to Humans
- PMID: 35336827
- PMCID: PMC8945790
- DOI: 10.3390/biology11030454
Advanced Diffusion MRI of the Visual System in Glaucoma: From Experimental Animal Models to Humans
Abstract
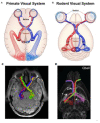
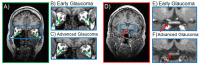
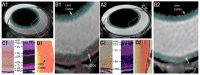
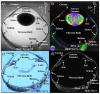
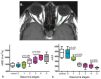
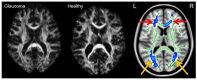
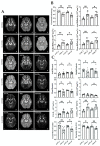
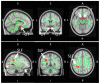
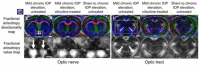
Glaucoma is a group of ophthalmologic conditions characterized by progressive retinal ganglion cell death, optic nerve degeneration, and irreversible vision loss. While intraocular pressure is the only clinically modifiable risk factor, glaucoma may continue to progress at controlled intraocular pressure, indicating other major factors in contributing to the disease mechanisms. Recent studies demonstrated the feasibility of advanced diffusion magnetic resonance imaging (dMRI) in visualizing the microstructural integrity of the visual system, opening new possibilities for non-invasive characterization of glaucomatous brain changes for guiding earlier and targeted intervention besides intraocular pressure lowering. In this review, we discuss dMRI methods currently used in visual system investigations, focusing on the eye, optic nerve, optic tract, subcortical visual brain nuclei, optic radiations, and visual cortex. We evaluate how conventional diffusion tensor imaging, higher-order diffusion kurtosis imaging, and other extended dMRI techniques can assess the neuronal and glial integrity of the visual system in both humans and experimental animal models of glaucoma, among other optic neuropathies or neurodegenerative diseases. We also compare the pros and cons of these methods against other imaging modalities. A growing body of dMRI research indicates that this modality holds promise in characterizing early glaucomatous changes in the visual system, determining the disease severity, and identifying potential neurotherapeutic targets, offering more options to slow glaucoma progression and to reduce the prevalence of this world's leading cause of irreversible but preventable blindness.
Keywords: diffusion; eye; glaucoma; magnetic resonance imaging; optic nerve; visual pathway.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

