Structural Insight of New Butyrylcholinesterase Inhibitors Based on Benzylbenzofuran Scaffold
- PMID: 35337102
- PMCID: PMC8955773
- DOI: 10.3390/ph15030304
Structural Insight of New Butyrylcholinesterase Inhibitors Based on Benzylbenzofuran Scaffold
Abstract
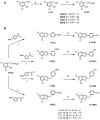
In the present work, we use a merger of computational and biochemical techniques as a rational guideline for structural modification of benzofuran derivatives to find pertinent structural features for the butyrylcholinesterase inhibitory activity and selectivity. Previously, we revealed a series of 2-phenylbenzofuran compounds that displayed a selective inhibitory activity for BChE. Here, in an effort to discover novel selective BChE inhibitors with favorable physicochemical and pharmacokinetic profiles, 2-benzylbenzofurans were designed, synthesized, and evaluated as BChE inhibitors. The 2-phenylbenzofuran scaffold structure is modified by introducing one methylene spacer between the benzofuran core and the 2-phenyl ring with a hydroxyl substituent in the para or meta position. Either position 5 or 7 of the benzofuran scaffold was substituted with a bromine or chlorine atom. Further assessment of the selected list of compounds indicated that the substituent's nature and position determined their activity and selectivity. 5-bromo-2-(4-hydroxybenzyl)benzofuran 9B proved to be the most potent butyrylcholinesterase inhibitor (IC50 = 2.93 µM) of the studied series. Computational studies were carried out to correlate the theoretical and experimental binding affinity of the compounds to the BChE protein.
Keywords: benzylbenzofuran; butyrylcholinesterase inhibitors; docking studies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
LinkOut - more resources
Full Text Sources
Miscellaneous

