Bona Fide Th17 Cells without Th1 Functional Plasticity Protect against Influenza
- PMID: 35338093
- PMCID: PMC9012674
- DOI: 10.4049/jimmunol.2100801
Bona Fide Th17 Cells without Th1 Functional Plasticity Protect against Influenza
Abstract
Optimal transcriptional programming needed for CD4 T cells to protect against influenza A virus (IAV) is unclear. Most IAV-primed CD4 T cells fit Th1 criteria. However, cells deficient for the Th1 "master regulator," T-bet, although marked by reduced Th1 identity, retain robust protective capacity. In this study, we show that T-bet's paralog, Eomesodermin (Eomes), is largely redundant in the presence of T-bet but is essential for the residual Th1 attributes of T-bet-deficient cells. Cells lacking both T-bet and Eomes instead develop concurrent Th17 and Th2 responses driven by specific inflammatory signals in the infected lung. Furthermore, the transfer of T-bet- and Eomes-deficient Th17, but not Th2, effector cells protects mice from lethal IAV infection. Importantly, these polyfunctional Th17 effectors do not display functional plasticity in vivo promoting gain of Th1 attributes seen in wild-type Th17 cells, which has clouded evaluation of the protective nature of Th17 programming in many studies. Finally, we show that primary and heterosubtypic IAV challenge is efficiently cleared in T-bet- and Eomes double-deficient mice without enhanced morbidity despite a strongly Th17-biased inflammatory response. Our studies thus demonstrate unexpectedly potent antiviral capacity of unadulterated Th17 responses against IAV, with important implications for vaccine design.
Copyright © 2022 by The American Association of Immunologists, Inc.
Figures



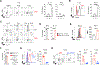
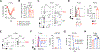

Similar articles
-
STAT1 Controls the Functionality of Influenza-Primed CD4 T Cells but Therapeutic STAT4 Engagement Maximizes Their Antiviral Impact.J Immunol. 2023 May 1;210(9):1292-1304. doi: 10.4049/jimmunol.2200407. J Immunol. 2023. PMID: 36961447 Free PMC article.
-
T-cell immunity against influenza virus does not require Th1 or Th17 master regulator transcription factors.Mucosal Immunol. 2025 Aug 15:S1933-0219(25)00088-1. doi: 10.1016/j.mucimm.2025.08.005. Online ahead of print. Mucosal Immunol. 2025. PMID: 40819821
-
Eomesodermin Expression in CD4+ T Cells Restricts Peripheral Foxp3 Induction.J Immunol. 2015 Nov 15;195(10):4742-52. doi: 10.4049/jimmunol.1501159. Epub 2015 Oct 9. J Immunol. 2015. PMID: 26453746
-
Ex Pluribus Unum: The CD4 T Cell Response against Influenza A Virus.Cells. 2024 Apr 5;13(7):639. doi: 10.3390/cells13070639. Cells. 2024. PMID: 38607077 Free PMC article. Review.
-
A cellular and molecular view of T helper 17 cell plasticity in autoimmunity.J Autoimmun. 2018 Feb;87:1-15. doi: 10.1016/j.jaut.2017.12.007. Epub 2017 Dec 22. J Autoimmun. 2018. PMID: 29275836 Review.
Cited by
-
Regulation of CD4 T Cell Responses by the Transcription Factor Eomesodermin.Biomolecules. 2022 Oct 24;12(11):1549. doi: 10.3390/biom12111549. Biomolecules. 2022. PMID: 36358898 Free PMC article. Review.
-
Preexisting vaccine-primed heterosubtypic T cell immunity protects the maternal-fetal unit from adverse influenza outcomes in mice.J Clin Invest. 2025 Jan 2;135(1):e179230. doi: 10.1172/JCI179230. J Clin Invest. 2025. PMID: 39744951 Free PMC article.
-
Recent Advances in the Development of Mincle-Targeting Vaccine Adjuvants.Vaccines (Basel). 2024 Nov 26;12(12):1320. doi: 10.3390/vaccines12121320. Vaccines (Basel). 2024. PMID: 39771982 Free PMC article. Review.
-
Cell-Cultured Influenza Vaccine Enhances IFN-γ+ T Cell and Memory T Cell Responses Following A/Victoria/2570/2019 IVR-215 (A/H1N1) Infection.Vaccines (Basel). 2024 Dec 11;12(12):1392. doi: 10.3390/vaccines12121392. Vaccines (Basel). 2024. PMID: 39772053 Free PMC article.
-
Diversifying T-cell responses: safeguarding against pandemic influenza with mosaic nucleoprotein.J Virol. 2025 Mar 18;99(3):e0086724. doi: 10.1128/jvi.00867-24. Epub 2025 Feb 3. J Virol. 2025. PMID: 39898643 Free PMC article.
References
-
- Liu B, Bao L, Wang L, Li F, Wen M, Li H, Deng W, Zhang X, and Cao B 2019. Anti-IFN-gamma therapy alleviates acute lung injury induced by severe influenza A (H1N1) pdm09 infection in mice. J Microbiol Immunol Infect - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

