ATP7B gene therapy of autologous reprogrammed hepatocytes alleviates copper accumulation in a mouse model of Wilson's disease
- PMID: 35340061
- PMCID: PMC9790736
- DOI: 10.1002/hep.32484
ATP7B gene therapy of autologous reprogrammed hepatocytes alleviates copper accumulation in a mouse model of Wilson's disease
Abstract
Background and aims: Wilson's disease (WD) is a rare hereditary disorder due to ATP7B gene mutation, causing pathologic copper storage mainly in the liver and neurological systems. Hepatocyte transplantation showed therapeutic potential; however, this strategy is often hindered by a shortage of quality donor cells and by allogeneic immune rejection. In this study, we aimed to evaluate the function and efficacy of autologous reprogrammed, ATP7B gene-restored hepatocytes using a mouse model of WD.
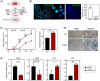
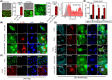
Approach and results: Sufficient liver progenitor cells (LPCs) were harvested by reprogramming hepatocytes from ATP7B-/- mice with small molecules, which exhibited strong proliferation and hepatic differentiation capacity in vitro. After lentivirus-mediated mini ATP7B gene transfection and redifferentiation, functional LPC-ATP7B-derived hepatocytes (LPC-ATP7B-Heps) were developed. RNA sequencing data showed that, compared with LPC-green fluorescent protein-Heps (LPC-GFP-Heps) with enrichment of genes that were mainly in pathways of oxidative stress and cell apoptosis, in LPC-ATP7B-Heps under high copper stress, copper ion binding and cell proliferation pathways were enriched. LPC-ATP7B-Heps transplantation into ATP7B-/- mice alleviated deposition of excess liver copper with its associated inflammation and fibrosis, comparable with those observed using normal primary hepatocytes at 4 months after transplantation.
Conclusions: We established a system of autologous reprogrammed WD hepatocytes and achieved ATP7B gene therapy in vitro. LPC-ATP7B-Heps transplantation demonstrated therapeutic efficacy on copper homeostasis in a mouse model of WD.
© 2022 The Authors. Hepatology published by Wiley Periodicals LLC on behalf of American Association for the Study of Liver Diseases.
Conflict of interest statement
Nothing to report.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

