Safety and immunogenicity of 3 formulations of a Sabin inactivated poliovirus vaccine produced on the PER.C6® cell line: A phase 2, double-blind, randomized, controlled study in infants vaccinated at 6, 10 and 14 weeks of age
- PMID: 35344464
- PMCID: PMC9196784
- DOI: 10.1080/21645515.2022.2044255
Safety and immunogenicity of 3 formulations of a Sabin inactivated poliovirus vaccine produced on the PER.C6® cell line: A phase 2, double-blind, randomized, controlled study in infants vaccinated at 6, 10 and 14 weeks of age
Abstract
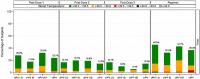
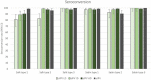
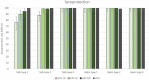
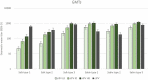
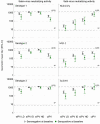
An inactivated poliovirus vaccine candidate using Sabin strains (sIPV) grown on the PER.C6® cell line was assessed in infants after demonstrated immunogenicity and safety in adults. The study recruited 300 infants who were randomized (1:1:1:1) to receive one of 3 dose levels of sIPV or a conventional IPV based on Salk strains (cIPV). Poliovirus-neutralizing antibodies were measured before the first dose and 28 days after the third dose. Reactogenicity was assessed for 7 days and unsolicited adverse events (AEs) for 28 days after each vaccination. Serious AEs (SAEs) were recorded throughout the study. Solicited AEs were mostly mild to moderate. None of the SAEs reported in the study were judged vaccine related, including one fatal SAE due to aspiration of vomitus that occurred 26 days after the third dose of low-dose sIPV. After 3 sIPV vaccinations and across all dose levels, seroconversion (SC) rates were at least 92% against Sabin poliovirus types and at least 80% against Salk types, with a dose-response in neutralizing antibody geometric mean titers (GMTs) observed across the 3 sIPV groups. Compared to cIPV, the 3 sIPV groups displayed similar or higher SC rates and GMTs against the 3 Sabin types but showed a lower response against Salk types 1 and 2; this was most visible for Salk type 1. While the PER.C6® cell line-based sIPV showed an acceptable safety profile and immunogenicity in infants, lower seroprotection against type 1 warrants optimization of dose level and additional clinical evaluation.
Keywords: IPV; Inactivated; PER.C6® cell line; Sabin; infants; poliovirus; vaccine.
Conflict of interest statement
G.Sh., A.G., M.Du., G.Sc., V.v.P., M.L.G., H.S. and M.Do. are full-time employees of the study sponsor. C.P.C. was a full-time employee of the sponsor at the time of the trial, J.-M. J. is an independent consultant under contract with the study sponsor. A.L.O.-L, M.T.-A., and D.C.-Y. have received grants from Janssen Vaccines to finance the conduct of this study in their institution. G.Sh, G.Sc, M.L.G., H.S., and A.G hold shares in Johnson & Johnson.
Figures
Similar articles
-
Safety and immunogenicity of a new Sabin inactivated poliovirus vaccine candidate produced on the PER.C6® cell-line: a phase 1 randomized controlled trial in adults.Hum Vaccin Immunother. 2021 May 4;17(5):1366-1373. doi: 10.1080/21645515.2020.1812315. Epub 2020 Nov 11. Hum Vaccin Immunother. 2021. PMID: 33175637 Free PMC article. Clinical Trial.
-
Safety and immunogenicity of experimental stand-alone trivalent, inactivated Sabin-strain polio vaccine formulations in healthy infants: A randomized, observer-blind, controlled phase 1/2 trial.Vaccine. 2020 Jul 14;38(33):5313-5323. doi: 10.1016/j.vaccine.2020.05.081. Epub 2020 Jun 17. Vaccine. 2020. PMID: 32563609 Free PMC article. Clinical Trial.
-
Immunogenicity of three sequential schedules with Sabin inactivated poliovirus vaccine and bivalent oral poliovirus vaccine in Zhejiang, China: an open-label, randomised, controlled trial.Lancet Infect Dis. 2020 Sep;20(9):1071-1079. doi: 10.1016/S1473-3099(19)30738-8. Epub 2020 May 19. Lancet Infect Dis. 2020. PMID: 32442523 Clinical Trial.
-
Immunogenicity of sequential inactivated and oral poliovirus vaccines (OPV) versus inactivated poliovirus vaccine (IPV) alone in healthy infants: A systematic review and meta-analysis.Hum Vaccin Immunother. 2018;14(11):2636-2643. doi: 10.1080/21645515.2018.1489188. Epub 2018 Jul 16. Hum Vaccin Immunother. 2018. PMID: 29985751 Free PMC article.
-
Current and next-generation formulation strategies for inactivated polio vaccines to lower costs, increase coverage, and facilitate polio eradication.Hum Vaccin Immunother. 2022 Dec 30;18(7):2154100. doi: 10.1080/21645515.2022.2154100. Epub 2022 Dec 28. Hum Vaccin Immunother. 2022. PMID: 36576132 Free PMC article. Review.
Cited by
-
A highly immunogenic UVC inactivated Sabin based polio vaccine.NPJ Vaccines. 2024 Nov 14;9(1):217. doi: 10.1038/s41541-024-00995-w. NPJ Vaccines. 2024. PMID: 39543143 Free PMC article.
-
Analysis of a Sabin-Strain Inactivated Poliovirus Vaccine Response to a Circulating Type 2 Vaccine-Derived Poliovirus Event in Sichuan Province, China 2019-2021.JAMA Netw Open. 2023 Jan 3;6(1):e2249710. doi: 10.1001/jamanetworkopen.2022.49710. JAMA Netw Open. 2023. PMID: 36602797 Free PMC article.
-
Immune and Safety Analysis of ultraIPVTM, a Novel UVC-Inactivated Polio Vaccine.Viruses. 2025 Jun 27;17(7):915. doi: 10.3390/v17070915. Viruses. 2025. PMID: 40733533 Free PMC article.
References
-
- Global Polio Eradication Initiative, World Health Organization. Polio endgame strategy 2019-2023. Eradication, integration, certification and containment. [accessed 2021 June 30]. http://polioeradication.org/wp-content/uploads/2019/06/english-polio-end....
-
- World Health Organization . Polio vaccines: WHO position paper – March, 2016. Wkly Epidemiol Rec. 2016;91(12):145–11. - PubMed
-
- Global Polio Eradication Initiative . GPEI polio today WPV reports. [accessed 2021 June 30]. https://polioeradication.org/polio-today/polio-now/this-week/.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
