A biomechanical model for evaluating the performance of accommodative intraocular lenses
- PMID: 35344827
- PMCID: PMC9119028
- DOI: 10.1016/j.jbiomech.2022.111054
A biomechanical model for evaluating the performance of accommodative intraocular lenses
Abstract
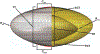
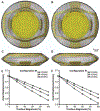
Accommodation alters the shape of the eye lens to change focus from distant to near vision. This function declines with age in the development of presbyopia and most people experience a near total loss of accommodative ability by 55 years. Currently, there are no surgical procedures that correct presbyopia, but considerable work has been done in the development of accommodative intraocular lenses (AIOLs) implanted during cataract surgery. Despite these efforts, AIOLs only restore ∼ 20% of youthful accommodative amplitude and they suffer from high rates of visually-debilitating fibrosis. An important design tool that is lacking that could aid in improving AIOL designs is modeling. Herein, we addressed this need through the development of a fully 3-D finite element model that was used to predict the behavior of a dual-optic AIOL implanted within the post-surgical lens capsule. Models of the native human lens were developed to identify the stress-free configuration of the lens capsule needed to accurately predict the accommodated state of the lens and the configuration of the zonular traction needed for the disaccommodated state. The AIOL model demonstrated the functional importance of implant stiffness and predicted an approximately linear relationship between zonular traction magnitude and axial displacement of the optics. To our knowledge, this is the first model that can be used to gain insights into AIOL efficacy. It provides a foundation for continued development of a predictive tool that could ultimately improve AIOL designs that seek to restore youthful accommodative function.
Keywords: Accommodation; Biomechanics; Computational modeling; Hyperelastic; Intraocular lens; Lens capsule.
Copyright © 2022 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Conflict of Interest
None of the authors have any professional or financial conflicts of interest.
Figures




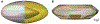
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

