Effect of LRRK2 protein and activity on stimulated cytokines in human monocytes and macrophages
- PMID: 35347144
- PMCID: PMC8960803
- DOI: 10.1038/s41531-022-00297-9
Effect of LRRK2 protein and activity on stimulated cytokines in human monocytes and macrophages
Abstract
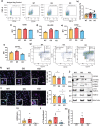
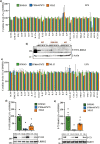
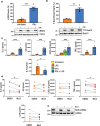
Leucine-rich-repeat kinase 2 (LRRK2), a potential therapeutic target for the treatment of Parkinson's disease (PD), is highly expressed in monocytes and macrophages and may play a role in the regulation of inflammatory pathways. To determine how LRRK2 protein levels and/or its activity modulate inflammatory cytokine/chemokine levels in human immune cells, isogenic human induced pluripotent stem cells (iPSC) with the LRRK2-activating G2019S mutation, wild-type LRRK2, and iPSC deficient in LRRK2 were differentiated to monocytes and macrophages and stimulated with inflammatory toll-like receptor (TLR) agonists in the presence and absence of LRRK2 kinase inhibitors. The effect of LRRK2 inhibitors and the effect of increasing LRRK2 levels with interferon gamma on TLR-stimulated cytokines were also assessed in primary peripheral blood-derived monocytes. Monocytes and macrophages with the LRRK2 G2019S mutation had significantly higher levels of cytokines and chemokines in tissue culture media following stimulation with TLR agonists compared to isogenic controls. Knockout of LRRK2 impaired phagocytosis but did not significantly affect TLR-mediated cytokine levels. Interferon gamma significantly increased the levels of LRRK2 and phosphorylation of its downstream Rab10 substrate, and potentiated TLR-mediated cytokine levels. LRRK2 kinase inhibitors did not have a major effect on TLR-stimulated cytokine levels. Results suggest that the LRRK2 G2019S mutation may potentiate inflammation following activation of TLRs. However, this was not dependent on LRRK2 kinase activity. Indeed, LRRK2 kinase inhibitors had little effect on TLR-mediated inflammation under the conditions employed in this study.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- Gasser T. Molecular pathogenesis of Parkinson disease: insights from genetic studies. Expert Rev. Mol. Med. 2009;11:e22. - PubMed
-
- Sheng Z, et al. Ser1292 autophosphorylation is an indicator of LRRK2 kinase activity and contributes to the cellular effects of PD mutations. Sci. Transl. Med. 2012;4:164ra161. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

