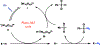Decatungstate-photocatalysed C(sp3)-H azidation
- PMID: 35348566
- PMCID: PMC9020444
- DOI: 10.1039/d2cc00425a
Decatungstate-photocatalysed C(sp3)-H azidation
Abstract
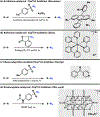
C-H Azidation is an increasingly important tool for bioconjugation, materials chemistry, and the synthesis of nitrogen-containing natural products. While several approaches have been developed, these often require exotic and energetic reagents, expensive photocatalysts, or both. Here we report a simple and general C-H azidation reaction using earth-abundant tetra-n-butylammonium decatungstate as a photocatalyst and commercial p-acetamidobenzenesulfonyl azide (p-ABSA) as the azide source. This system can azidate a variety of unactivated C(sp3)-H bonds in moderate to good yields and excellent turnover numbers. Preliminary mechanistic experiments implicate a radical mechanism proceeding VIA photo-hydrogen atom transfer (photo-HAT).
Conflict of interest statement
Conflicts of interest
There are no conflicts to declare.
Figures
Similar articles
-
Simple, catalytic C(sp3)-H azidation using the C-H donor as the limiting reagent.Chem Commun (Camb). 2024 Mar 28;60(27):3705-3708. doi: 10.1039/d3cc04728h. Chem Commun (Camb). 2024. PMID: 38477139 Free PMC article.
-
Metal-catalysed azidation of tertiary C-H bonds suitable for late-stage functionalization.Nature. 2015 Jan 29;517(7536):600-4. doi: 10.1038/nature14127. Nature. 2015. PMID: 25631448 Free PMC article.
-
Remote Radical Azidation of Unactivated C(sp3)-H Bonds in Sulfamoyl Azides.Org Lett. 2024 May 3;26(17):3519-3523. doi: 10.1021/acs.orglett.4c00862. Epub 2024 Apr 23. Org Lett. 2024. PMID: 38651932
-
Chemical versatility of azide radical: journey from a transient species to synthetic accessibility in organic transformations.Chem Soc Rev. 2022 Mar 21;51(6):2255-2312. doi: 10.1039/d1cs00494h. Chem Soc Rev. 2022. PMID: 35229836 Review.
-
Reactivities of vinyl azides and their recent applications in nitrogen heterocycle synthesis.Org Biomol Chem. 2015 Apr 7;13(13):3844-55. doi: 10.1039/c5ob00099h. Org Biomol Chem. 2015. PMID: 25731154 Free PMC article. Review.
Cited by
-
Simple, catalytic C(sp3)-H azidation using the C-H donor as the limiting reagent.Chem Commun (Camb). 2024 Mar 28;60(27):3705-3708. doi: 10.1039/d3cc04728h. Chem Commun (Camb). 2024. PMID: 38477139 Free PMC article.
-
Building Catalytic Reactions One Electron at a Time.Acc Chem Res. 2024 Oct 15;57(20):3068-3078. doi: 10.1021/acs.accounts.4c00515. Epub 2024 Sep 24. Acc Chem Res. 2024. PMID: 39317431
References
-
- Bräse S, Gil C, Knepper K and Zimmermann V, Angew. Chem. Int. Ed, 2005, 44, 5188 – 5240. - PubMed
- Jewett JC and Bertozzi CR, Chem. Soc. Rev, 2010, 39, 1272 – 1279. - PMC - PubMed
- Thirumurugan P, Matosiuk D and Jozwiak K, Chem. Rev, 2013, 113, 4905 – 4979. - PubMed
- Tron GC, Pirali T, Billington RA, Canonico PL, Sorba G and Genazzani AA, Med. Res. Rev, 2008, 28, 278 – 308. - PubMed
- Tanimoto H and Kakiuchi K, Nat. Prod. Commun, 2013, 8, 1021 – 1034. - PubMed
- Tuktarov AR, Akhmetov AR and Dzhemilev UM, Mater. Sci. Res. J, 2014, 8, 123 – 166.
- Schilling CI, Jung N, Biskup M, Schepers U and Bräse S, Chem. Soc. Rev, 2011, 40, 4840 – 4871. - PubMed
- Lutz J-F and Zarafshani Z, Adv. Drug Deliv. Rev, 2008, 60, 958 – 970. - PubMed
-
- For selected applications of azide to synthesize bioactive molecules, see: Balci M, Synthesis, 2018, 50, 1373 – 1401.
- Baran PS and Zografos AL, O’Malley DP, J. Am. Chem. Soc, 2004, 126, 3726 – 3727. - PubMed
- Tanaka H, Sawayama AM and Wandless TJ, J. Am. Chem. Soc, 2003, 125, 6864 – 6865. - PubMed
- Fuchs JR and Funk RL, J. Am. Chem. Soc, 2004, 126, 5068 – 5069. - PubMed
- Wrobleski A, Sahasrabudhe K and Aubé J, J. Am. Chem. Soc, 2004, 126, 5475 – 5481. - PubMed
- Zwick CR III and Renata H, J. Am. Chem. Soc, 2018, 140, 1165 – 1169. - PubMed
-
- For selected examples of azidation reagent, see: Alazet S, Preindl J, Simonet-Davin R, Nicolai S, Nanchen A, Meyer T and Waser J, J. Org. Chem, 2018, 83, 12334 – 12356. - PubMed
- Zhdankin VV, Kuehl CJ, Krasutsky AP, Formaneck MS and Bolz JT, Tetrahedron Lett, 1994, 35, 9677 – 9680.
- Zhdankin VV, Krasutsky AP, Kuehl CJ, Simonsen AJ, Woodward JW, Mismash B and Bolz JT, J. Am. Chem. Soc, 1996, 118, 5192 – 5197.
- Breslow DS, Sloan MF, Newburg NR and Renfrow WB, J. Am. Chem. Soc, 1969, 91, 2273 – 2279.
- Goddard-Borger ED and Stick RV, Org. Lett, 2007, 9, 3797 –3800. - PubMed
-
- Magnus P and Lacour J, J. Am. Chem. Soc,1992, 114, 767 – 769.
- Kita Y, Tohma H, Takada T, Mitoh S, Fujita S and Gyoten M, Synlett, 1994, 6, 427 – 428.
- Magnus P, Lacour J and Weber W, J. Am. Chem. Soc, 1993, 115, 9347 –9348.
- Magnus P, Hulme C and Weber W, J. Am. Chem. Soc, 1994, 116, 4501 – 4502.
- Magnus P, Lacour J, Evans PA, Roe MB and Hulme C, J. Am. Chem. Soc, 1996, 118, 3406 – 3418.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources