Computational analysis of cortical neuronal excitotoxicity in a large animal model of neonatal brain injury
- PMID: 35351004
- PMCID: PMC8966144
- DOI: 10.1186/s11689-022-09431-3
Computational analysis of cortical neuronal excitotoxicity in a large animal model of neonatal brain injury
Abstract
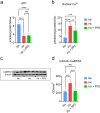
Background: Neonatal hypoxic brain injury is a major cause of intellectual and developmental disability. Hypoxia causes neuronal dysfunction and death in the developing cerebral cortex due to excitotoxic Ca2+-influx. In the translational piglet model of hypoxic encephalopathy, we have previously shown that hypoxia overactivates Ca2+/Calmodulin (CaM) signaling via Sarcoma (Src) kinase in cortical neurons, resulting in overexpression of proapoptotic genes. However, identifying the exact relationship between alterations in neuronal Ca2+-influx, molecular determinants of cell death, and the degree of hypoxia in a dynamic system represents a significant challenge.
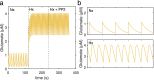
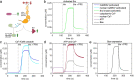
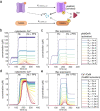
Methods: We used experimental and computational methods to identify molecular events critical to the onset of excitotoxicity-induced apoptosis in the cerebral cortex of newborn piglets. We used 2-3-day-old piglets (normoxic [Nx], hypoxic [Hx], and hypoxic + Src-inhibitor-treatment [Hx+PP2] groups) for biochemical analysis of ATP production, Ca2+-influx, and Ca2+/CaM-dependent protein kinase kinase 2 (CaMKK2) expression. We then used SimBiology to build a computational model of the Ca2+/CaM-Src-kinase signaling cascade, simulating Nx, Hx, and Hx+PP2 conditions. To evaluate our model, we used Sobol variance decomposition, multiparametric global sensitivity analysis, and parameter scanning.
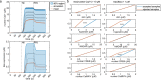
Results: Our model captures important molecular trends caused by hypoxia in the piglet brain. Incorporating the action of Src kinase inhibitor PP2 further validated our model and enabled predictive analysis of the effect of hypoxia on CaMKK2. We determined the impact of a feedback loop related to Src phosphorylation of NMDA receptors and activation kinetics of CaMKII. We also identified distinct modes of signaling wherein Ca2+ level alterations following Src kinase inhibition may not be a linear predictor of changes in Bax expression. Importantly, our model indicates that while pharmacological pre-treatment significantly reduces the onset of abnormal Ca2+-influx, there exists a window of intervention after hypoxia during which targeted modulation of Src-NMDAR interaction kinetics in combination with PP2 administration can reduce Ca2+-influx and Bax expression to similar levels as pre-treatment.
Conclusions: Our model identifies new dynamics of critical components in the Ca2+/CaM-Src signaling pathway leading to neuronal injury and provides a feasible framework for drug efficacy studies in translational models of neonatal brain injury for the prevention of intellectual and developmental disabilities.
Keywords: Calcium/calmodulin; Computational modeling; Excitotoxicity; Neonatal brain injury; Nuclear calcium; SimBiology; Src kinase.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Burd I, Welling J, Kannan G, Johnston M, v. Excitotoxicity as a common mechanism for fetal neuronal injury with hypoxia and intrauterine inflammation. Adv Pharmacol. 2016;76:85–101. - PubMed
-
- Lawn JE, Cousens S, Zupan J. 4 million neonatal deaths: When? Where? Why? Lancet. 2005;365:891–900. - PubMed
-
- Rossi DJ, Oshima T, Attwell D. Glutamate release in severe brain ischaemia is mainly by reversed uptake. Nature. 2000;403(6767):316–321. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

