The effect of gluten in adolescents and young adults with gastrointestinal symptoms: a blinded randomised cross-over trial
- PMID: 35352373
- PMCID: PMC9313792
- DOI: 10.1111/apt.16914
The effect of gluten in adolescents and young adults with gastrointestinal symptoms: a blinded randomised cross-over trial
Abstract
Background: The popularity of the gluten-free diet and sales of gluten-free products have increased immensely.
Aims: To investigate whether gluten induces gastrointestinal symptoms, measured by self-reported questionnaires, as well as mental health symptoms in adolescents from a population-based cohort.
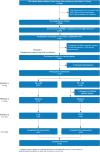
Methods: The eligible participants (n = 273) were recruited from a population-based cohort of 1266 adolescents and had at least four different gastrointestinal symptoms. Phase one (n = 54) was a run-in phase where the participants lived gluten-free for 2 weeks. If they improved they continued to phase 2 (n = 33), a blinded randomised cross-over trial. Participants were blindly randomised either to start with 7 days of gluten, eating two granola bars containing 10 g of gluten or to 7 days on placebo, eating two granola bars without gluten, followed by the reverse and separated by a 7-day washout period. The effects of the intervention on gastrointestinal symptoms and mental health symptoms were assessed.
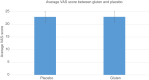
Results: In total, 54/273 participants entered the run-in phase and 35 were eligible for randomization. A total of 33 were randomised and 32 completed the trial. The median age was 20.3 (IQR 19.2-20.9) and 32/33 participants were females. Compared with a placebo, gluten did not induce gastrointestinal symptoms. The difference in the average VAS was -0.01 (95% confidence interval -2.07 to 2.05). Nor did we find a difference in the outcomes measuring mental health.
Conclusion: Compared with placebo, adding gluten to the diet did not induce gastrointestinal symptoms or worsened mental health in adolescents recruited from a population-based cohort. The trial registration number is NCT04639921.
Keywords: gluten; gluten sensitivity enteropathy; gluten-free diet.
© 2022 The Authors. Alimentary Pharmacology & Therapeutics published by John Wiley & Sons Ltd.
Figures
Comment in
-
Editorial: lack of gastrointestinal symptoms caused by gluten in patients without coeliac disease-time to ditch the 'gluten' from 'non-coeliac gluten sensitivity'.Aliment Pharmacol Ther. 2022 Jul;56(2):340-341. doi: 10.1111/apt.16951. Aliment Pharmacol Ther. 2022. PMID: 35748828 No abstract available.
-
Letter: progressive weakening of the concept that gluten has a detrimental effect on mental health and gut symptoms in the absence of coeliac disease-authors' reply.Aliment Pharmacol Ther. 2022 Jul;56(2):365. doi: 10.1111/apt.17051. Aliment Pharmacol Ther. 2022. PMID: 35748833 No abstract available.
-
Editorial: lack of gastrointestinal symptoms caused by gluten in patients without coeliac disease-time to ditch the 'gluten' from 'non-coeliac gluten sensitivity'. Authors' reply.Aliment Pharmacol Ther. 2022 Jul;56(2):342. doi: 10.1111/apt.16980. Aliment Pharmacol Ther. 2022. PMID: 35748842 No abstract available.
-
Letter: progressive weakening of the concept that gluten has a detrimental effect on mental health and gut symptoms in the absence of coeliac disease.Aliment Pharmacol Ther. 2022 Jul;56(2):363-364. doi: 10.1111/apt.16963. Aliment Pharmacol Ther. 2022. PMID: 35748844 No abstract available.
References
-
- Perrin L, Allès B, Buscail C, Ravel C, Hercberg S, Julia C, et al. Gluten‐free diet in French adults without coeliac disease: sociodemographic characteristics, motives and dietary profile. Br J Nutr. 2019;122(2):231–9. - PubMed
-
- Potter MD, Jones MP, Walker MM, Koloski NA, Keely S, Holtmann G, et al. Incidence and prevalence of self‐reported non‐coeliac wheat sensitivity and gluten avoidance in Australia. Med J Australia. 2020;212(3):126–31. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
