Final analysis of the phase III non-inferiority COLUMBA study of subcutaneous versus intravenous daratumumab in patients with relapsed or refractory multiple myeloma
- PMID: 35354247
- PMCID: PMC9521240
- DOI: 10.3324/haematol.2021.279459
Final analysis of the phase III non-inferiority COLUMBA study of subcutaneous versus intravenous daratumumab in patients with relapsed or refractory multiple myeloma
Abstract
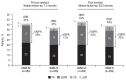
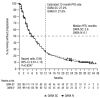
In the primary analysis of the phase III COLUMBA study, daratumumab by subcutaneous administration (DARA SC) demonstrated non-inferiority to intravenous administration (DARA IV) for relapsed or refractory multiple myeloma (RRMM). Here, we report the final analysis of efficacy and safety from COLUMBA after a median of 29.3 months follow-up (additional 21.8 months after the primary analysis). In total, 522 patients were randomized (DARA SC, n=263; DARA IV, n=259). With longer follow-up, DARA SC and DARA IV continued to show consistent efficacy and maximum trough daratumumab concentration as compared with the primary analysis. The overall response rate was 43.7% for DARA SC and 39.8% for DARA IV. The maximum mean (standard deviation [SD]) trough concentration (cycle 3, day 1 pre-dose) of serum DARA was 581 (SD, 315) μg/mL for DARA SC and 496 (SD, 231) μg/mL for DARA IV. Median progression-free survival was 5.6 months for DARA SC and 6.1 months for DARA IV; median overall survival was 28.2 months and 25.6 months, respectively. Grade 3/4 treatment-emergent adverse events occurred in 50.8% of patients in the DARA SC group and 52.7% in the DARA IV group; the most common (≥10%) were thrombocytopenia (DARA SC, 14.2%; DARA IV, 13.6%), anemia (13.8%; 15.1%), and neutropenia (13.1%; 7.8%). The safety profile remained consistent with the primary analysis after longer follow-up. In summary, DARA SC and DARA IV continue to demonstrate similar efficacy and safety, with a low rate of infusion-related reactions (12.7% vs. 34.5%, respectively) and shorter administration time (3-5 minutes vs. 3-7 hours) supporting DARA SC as a preferable therapeutic choice. (Clinicaltrials gov. Identifier: NCT03277105.
Figures



References
-
- de Weers M, Tai YT, van der Veer MS, et al. . Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other hematological tumors. J Immunol. 2011;186(3):1840-1848. - PubMed
-
- Lammerts van Bueren J, Jakobs D, Kaldenhoven N, et al. . Direct in vitro comparison of daratumumab with surrogate analogs of CD38 antibodies MOR03087, SAR650984 and Ab79. Blood. 2014;124(21):3474.
-
- Overdijk MB, Jansen JH, Nederend M, et al. . The therapeutic CD38 monoclonal antibody daratumumab induces programmed cell death via Fcγ receptor-mediated cross-linking. J Immunol. 2016;197(3):807-813. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

