National experience in the first two years of primary human papillomavirus (HPV) cervical screening in an HPV vaccinated population in Australia: observational study
- PMID: 35354610
- PMCID: PMC8965648
- DOI: 10.1136/bmj-2021-068582
National experience in the first two years of primary human papillomavirus (HPV) cervical screening in an HPV vaccinated population in Australia: observational study
Abstract
Objective: To review the first two years of the primary human papillomavirus (HPV) cervical screening programme in an HPV vaccinated population.
Design: Observational study.
Setting: Australia.
Participants: 3 745 318 women with a primary HPV test between 1 December 2017 and 31 December 2019; most women aged <40 years had previously been offered vaccination against HPV16 and HPV18.
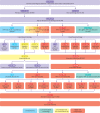
Interventions: Primary HPV screening with referral if HPV16 or HPV18 (HPV16/18) positive and triage with liquid based cytology testing (threshold atypical squamous cells-cannot exclude high grade squamous intraepithelial lesion) for women who were positive for high risk HPV types other than 16/18. A 12 month follow-up HPV test was recommended in triaged women with a negative or low grade cytology result, with referral if they tested positive for any high risk HPV type at follow-up.
Main outcome measures: Proportion of women who had attended for their first HPV screening test, tested positive, and were referred for colposcopy; and short term risk of detecting cervical intraepithelial neoplasia (CIN) grade 2 or worse, CIN grade 3 or worse, or cancer.
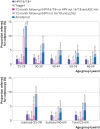
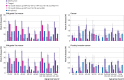
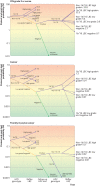
Results: 54.6% (n=3 507 281) of an estimated 6 428 677 eligible women aged 25-69 had undergone their first HPV test by the end of 2019. Among those attending for routine screening, positivity for HPV16/18 and for HPV types not 16/18 was, respectively, 2.0% and 6.6% in women aged 25-69 (n=3 045 844) and 2.2% and 13.3% in highly vaccinated cohorts of women aged 25-34 (n=768 362). Colposcopy referral (ages 25-69 years) was 3.5%, increasing to an estimated 6.2% after accounting for women who had not yet had a 12 month repeat test. Cervical cancer was detected in 0.98% (456/46 330) of women positive for HPV16/18 at baseline, including 0.32% (89/28 003) of women with HPV16/18 and negative cytology. Women with HPV types not 16/18 and negative or low grade cytology at both baseline and 12 months were at low risk of serious disease (3.4% CIN grade 3 or worse; 0.02% cancer; n=20 019) but estimated to account for 62.0% of referrals for this screening algorithm.
Conclusions: Colposcopy referral thresholds need to consider underlying cancer risk; on this basis, women with HPV16/18 in the first round of HPV screening were found to be at higher risk regardless of cytology result, even in a previously well screened population. Women with HPV types not 16/18 and negative or low grade cytology showed a low risk of serious abnormalities but constitute most referrals and could be managed safely with two rounds of repeat HPV testing rather than one. HPV16/18 driven referrals were low in HPV vaccinated cohorts.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: support from the National Health and Medical Research Council (salary support for MSm, MSh, and KC), Cancer Institute NSW (salary support for MSm), the Horizon 2020 Framework Programme for Research and Innovation of the European Commission, through the Risk-based Screening for Cervical Cancer Network (grant to MA), Cancer Council NSW (grant to Sciensano (employer of MA) to support a meta-analysis on triage of HPV positive women), and Commonwealth Department of Health (provision of aggregated data) for the current work; and the following relationships with organisations that might have an interest in the submitted work in the previous three years: National Health and Medical Research Council (grants to MSa and KC), Commonwealth Department of Health (contract funding for other work to MSm and KC), National Institutes of Health (grants to PC), Centers for Disease Control and Prevention (contracts to PC), Sequiris (honorariums to CDW for lectures and sponsorship for conference travel), Biogen (honorariums to CDW for lectures), and CSL (shares owned by CDW). KC and MSa are co-principal investigators of the investigator initiated trial of cytology and primary human papillomavirus (HPV) screening in Australia (Compass), which is coordinated at the Australian Centre for the Prevention of Cervical Cancer (ACPCC), a government funded not-for-profit health promotion charity. The ACPCC has received equipment and a funding contribution for the Compass trial from Roche Molecular Systems USA, and operational support for the Compass trial from the Australian government. MC, PC, and DW are also investigators on Compass. Neither KC nor MC, nor their institution on their behalf, have received direct or indirect funding from industry for Compass or any other project. MSm and MSh are involved in analysis of Compass data but otherwise declare they have no conflicts of interest. KC and MSa are also co-principal investigators on a major investigator initiated implementation program “Elimination of Cervical Cancer in the Western Pacific (ECCWP),” which will receive support from the Minderoo Foundation and the Frazer Family Foundation and equipment donations from Cepheid. PC has received HPV tests and assays at a reduced or no cost for research from Roche, Becton Dickinson, Cepheid, and Arbor Vita. DG has received personal payment from Johnson & Johnson for preparation of expert testimony, and serves on the Australian government National Quality and Safety Monitoring Committee and Victorian Regional Committee of the Australasian Faculty of Public Health Medicine. MSa is a director of Cancer Council Australia (unpaid role) and co-chair of Chair HPV Test Characteristics Expert Panel and Consultant (fees paid to Australian Centre for the Prevention of Cervical Cancer) and has received free testing kits for research from Roche, Seegene, Cepheid, Becton Dickinson, Abbott, AusDiagnostics, and Atila Biosystems. The opinions expressed by the authors are their own and this material should not be interpreted as representing the official viewpoint of the US Department of Health and Human Services, the National Institutes of Health, or the National Cancer Institute.
Figures
References
-
- Isidean SD, Mayrand MH, Ramanakumar AV, et al. CCCaST Study Group . Human papillomavirus testing versus cytology in primary cervical cancer screening: End-of-study and extended follow-up results from the Canadian cervical cancer screening trial. Int J Cancer 2016;139:2456-66. 10.1002/ijc.30385. - DOI - PubMed
