Regulatory Information and Guidance on Biosimilars and Their Use Across Europe: A Call for Strengthened One Voice Messaging
- PMID: 35355594
- PMCID: PMC8959407
- DOI: 10.3389/fmed.2022.820755
Regulatory Information and Guidance on Biosimilars and Their Use Across Europe: A Call for Strengthened One Voice Messaging
Abstract
Background: Beyond evaluation and approval, European and national regulators have a key role in providing reliable information on biosimilars and the science underpinning their development, approval, and use.
Objectives: This study aims to (i) review biosimilar information and guidance provided by EMA and national medicines agencies and (ii) explore stakeholder perspectives on the role of regulators in enabling acceptance and use of biosimilars.
Methods: This study consists of (i) a comparative review of regulatory information and position statements across medicine agencies (n = 32) and (ii) qualitative interviews with stakeholders in Europe (n = 14).
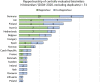
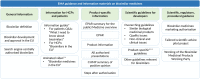
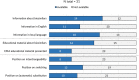
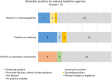
Results: The comparative analysis showed that regulatory information and guidance about biosimilars offered by national medicines agencies in Europe varies, and is limited or absent in multiple instances. Approximately 40% (13/31) of the national medicines agencies' websites did not offer any information regarding biosimilars, and for about half (15/31) no educational materials were provided. Only less than half of national medicines agencies provided guidance on biosimilar interchangeability and switching (8/31 and 12/31, respectively). Among the national medicines agencies that did offer guidance, the extent (e.g., elaborate position vs. brief statement) and content (e.g., full endorsement vs. more cautious) of the guidance differed substantially. Countries that have a strong involvement in EU level biosimilar regulatory activities generally had more elaborate information nationally. Interviewees underwrote the need for (national) regulators to intensify biosimilar stakeholder guidance, especially in terms of providing clear positions regarding biosimilar interchangeability and switching, which in turn can be disseminated by the relevant professional societies more locally.
Conclusion: This study revealed that, despite strong EU-level regulatory biosimilar guidance, guidance about biosimilars, and their use differs considerably across Member States. This heterogeneity, together with the absence of a clear EU-wide position on interchangeability, may instill uncertainty among stakeholders about the appropriate use of biosimilars in practice. Regulators should strive for a clear and common EU scientific position on biosimilar interchangeability to bridge this gap and unambiguously inform policy makers, healthcare professionals, and patients. Furthermore, there is a clear opportunity to expand information at the national level, and leverage EU-developed information materials more actively in this regard.
Keywords: biological; biosimilar; guidance; interchangeability; policy; regulatory; substitution; switching.
Copyright © 2022 Barbier, Mbuaki, Simoens, Declerck, Vulto and Huys.
Conflict of interest statement
IH, SS, PD, and AV are the founders of the KU Leuven Fund on Market Analysis of Biologics and Biosimilars following Loss of Exclusivity (MABEL Fund). AV is involved in consulting, educational work and speaking engagements for a number of companies, i.e., AbbVie, Accord, Amgen, Biogen, Effik, EGA, Pfizer/Hospira, Fresenius-Kabi, Mundipharma, Roche, Novartis, Sandoz, Boehringer Ingelheim. SS was involved in a stakeholder roundtable on biologics and biosimilars sponsored by Amgen, Pfizer, and MSD, and he has participated in advisory board meetings for Amgen, Pfizer, and Sandoz. He has contributed to studies on biologics and biosimilars for Hospira, Celltrion, Mundipharma, and Pfizer; and he has had speaking engagements for Amgen, Celltrion and Sandoz. PD participated at advisory board meetings for AbbVie, Amgen, Hospira, and Samsung Bioepis and is on the Speakers' Bureau of AbbVie, Celltrion, Hospira, Merck Serono, and Roche. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- European Medicines Agency . Biosimilars in the EU - Information guide for Healthcare Professionals (2017).
-
- European Medicines Agency . Guideline on Similar Biological Medicinal Products (2014).
-
- ICMRA . ICMRA Statement About Confidence in Biosimilar Products (for healthcare professionals) (2019).
-
- IQVIA . The Impact of Biosimilar Competition in Europe (2018).
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

