Electrophysiologic Conservation of Epicardial Conduction Dynamics After Myocardial Infarction and Natural Heart Regeneration in Newborn Piglets
- PMID: 35355973
- PMCID: PMC8959497
- DOI: 10.3389/fcvm.2022.829546
Electrophysiologic Conservation of Epicardial Conduction Dynamics After Myocardial Infarction and Natural Heart Regeneration in Newborn Piglets
Abstract
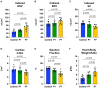
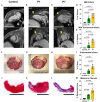
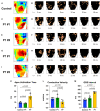
Newborn mammals, including piglets, exhibit natural heart regeneration after myocardial infarction (MI) on postnatal day 1 (P1), but this ability is lost by postnatal day 7 (P7). The electrophysiologic properties of this naturally regenerated myocardium have not been examined. We hypothesized that epicardial conduction is preserved after P1 MI in piglets. Yorkshire-Landrace piglets underwent left anterior descending coronary artery ligation at age P1 (n = 6) or P7 (n = 7), After 7 weeks, cardiac magnetic resonance imaging was performed with late gadolinium enhancement for analysis of fibrosis. Epicardial conduction mapping was performed using custom 3D-printed high-resolution mapping arrays. Age- and weight-matched healthy pigs served as controls (n = 6). At the study endpoint, left ventricular (LV) ejection fraction was similar for controls and P1 pigs (46.4 ± 3.0% vs. 40.3 ± 4.9%, p = 0.132), but significantly depressed for P7 pigs (30.2 ± 6.6%, p < 0.001 vs. control). The percentage of LV myocardial volume consisting of fibrotic scar was 1.0 ± 0.4% in controls, 9.9 ± 4.4% in P1 pigs (p = 0.002 vs. control), and 17.3 ± 4.6% in P7 pigs (p < 0.001 vs. control, p = 0.007 vs. P1). Isochrone activation maps and apex activation time were similar between controls and P1 pigs (9.4 ± 1.6 vs. 7.8 ± 0.9 ms, p = 0.649), but significantly prolonged in P7 pigs (21.3 ± 5.1 ms, p < 0.001 vs. control, p < 0.001 vs. P1). Conduction velocity was similar between controls and P1 pigs (1.0 ± 0.2 vs. 1.1 ± 0.4 mm/ms, p = 0.852), but slower in P7 pigs (0.7 ± 0.2 mm/ms, p = 0.129 vs. control, p = 0.052 vs. P1). Overall, our data suggest that epicardial conduction dynamics are conserved in the setting of natural heart regeneration in piglets after P1 MI.
Keywords: conduction; electrophysiology; heart; mapping; myocardial infarction; neonate - age; regeneration.
Copyright © 2022 Wang, Pong, Obafemi, Lucian, Aparicio-Valenzuela, Tran, Mullis, Elde, Tada, Baker, Wang, Cyr, Paulsen, Zhu, Lee and Woo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

