A Model of Human Small Airway on a Chip for Studies of Subacute Effects of Inhalation Toxicants
- PMID: 35357501
- PMCID: PMC9154286
- DOI: 10.1093/toxsci/kfac036
A Model of Human Small Airway on a Chip for Studies of Subacute Effects of Inhalation Toxicants
Abstract
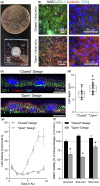
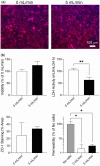
Testing for acute inhalation hazards is conducted in animals; however, a number of robust in vitro human cell-based alternatives have been developed and tested. These models range in complexity from cultures of cell lines or primary cells in air-liquid interface on Transwells, to more complex and physiologically relevant flow- and mechanical stimulation-enabled tissue chips. Although the former models are relatively straightforward to establish and can be tested in medium/high throughput, the latter require specialized equipment and lack in throughput. In this study, we developed a device that can be easily manufactured while allowing for the production of a differentiated lung tissue. This multilayered microfluidic device enables coculture of primary human small airway epithelial cells and lung microvascular endothelial cells under physiological conditions for up to 18 days and recreates the parenchymal-vascular interface in the distal lung. To explore the potential of this airway on a chip for applications in inhalation toxicology, we also devised a system that allows for direct gas/aerosol exposures of the engineered airway epithelium to noxious stimuli known to cause adverse respiratory effects, including dry flowing air, lipopolysaccharide, particulate matter, and iodomethane. This study generated quantitative, high-content data that were indicative of aberrant changes in biochemical (lactate dehydrogenase), barrier (dextran permeability), functional (ciliary beating), and molecular (imaging for various markers) phenotypes of the small airway epithelium due to inhalational exposures. This study is significant because it established an in vitro model of human small airway on a chip that can be used in medium/high-throughput studies of subacute effects of inhalation toxicants.
Keywords: in vitro models; hazard identification; lung.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Society of Toxicology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures



References
-
- Ainslie G. R., Davis M., Ewart L., Lieberman L. A., Rowlands D. J., Thorley A. J., Yoder G., Ryan A. M. (2019). Microphysiological lung models to evaluate the safety of new pharmaceutical modalities: A biopharmaceutical perspective. Lab Chip. 19, 3152–3161. - PubMed
-
- Avila A. M., Bebenek I., Bonzo J. A., Bourcier T., Davis Bruno K. L., Carlson D. B., Dubinion J., Elayan I., Harrouk W., Lee S. L., et al. (2020). An FDA/CDER perspective on nonclinical testing strategies: Classical toxicology approaches and new approach methodologies (NAMs). Regul. Toxicol. Pharmacol. 114, 104662. - PubMed
-
- Balogh Sivars K., Sivars U., Hornberg E., Zhang H., Branden L., Bonfante R., Huang S., Constant S., Robinson I., Betts C. J., et al. (2018). A 3D human airway model enables prediction of respiratory toxicity of inhaled drugs in vitro. Toxicol. Sci. 162, 301–308. - PubMed
-
- Benam K. H., Mazur M., Choe Y., Ferrante T. C., Novak R., Ingber D. E. (2017). Human lung small airway-on-a-chip protocol. Methods Mol. Biol. 1612, 345–365. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

