Chemogenetics of cell surface receptors: beyond genetic and pharmacological approaches
- PMID: 35359495
- PMCID: PMC8905536
- DOI: 10.1039/d1cb00195g
Chemogenetics of cell surface receptors: beyond genetic and pharmacological approaches
Abstract
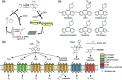
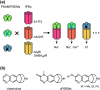
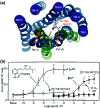
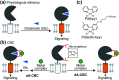
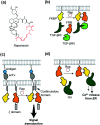
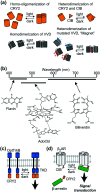
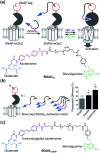
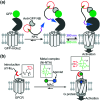
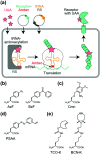
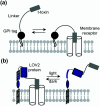
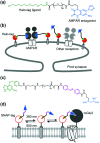
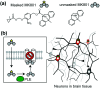
Cell surface receptors transmit extracellular information into cells. Spatiotemporal regulation of receptor signaling is crucial for cellular functions, and dysregulation of signaling causes various diseases. Thus, it is highly desired to control receptor functions with high spatial and/or temporal resolution. Conventionally, genetic engineering or chemical ligands have been used to control receptor functions in cells. As the alternative, chemogenetics has been proposed, in which target proteins are genetically engineered to interact with a designed chemical partner with high selectivity. The engineered receptor dissects the function of one receptor member among a highly homologous receptor family in a cell-specific manner. Notably, some chemogenetic strategies have been used to reveal the receptor signaling of target cells in living animals. In this review, we summarize the developing chemogenetic methods of transmembrane receptors for cell-specific regulation of receptor signaling. We also discuss the prospects of chemogenetics for clinical applications.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures






References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

