Molecular epidemiology and clinical characteristics of hepatitis D virus infection in Canada
- PMID: 35360523
- PMCID: PMC8961228
- DOI: 10.1016/j.jhepr.2022.100461
Molecular epidemiology and clinical characteristics of hepatitis D virus infection in Canada
Abstract
Background & aims: HDV affects 4.5-13% of chronic hepatitis B (CHB) patients globally, yet the prevalence of HDV infection in Canada is unknown. To investigate the prevalence, genotype, demographics, and clinical characteristics of HDV in Canada, we conducted a retrospective analysis of (1) HDV antibody and RNA positivity among referred specimens, and (2) a cross-sectional subset study of 135 HDV seropositive +/-RNA (HDV+) patients compared with 5,132 HBV mono-infected patients in the Canadian HBV Network.
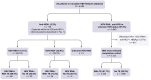
Methods: Anti-HDV IgG-positive specimens collected between 2012 and 2019 were RNA tested and the genotype determined. Patients enrolled in the Canadian HBV Network were >18 years of age and HBsAg-positive. Clinical data collected included risk factors, demographics, comorbidities, treatment, fibrosis assessment, and hepatic complications.
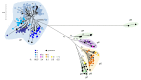
Results: Of the referred patients, 338/7,080 (4.8%, 95% CI 4.3-5.3) were HDV seropositive, with 219/338 RNA-positive (64.8%, 95% CI 59.6-69.7). The HDV+ cohort were more likely to be born in Canada, to be White or Black/African/Caribbean than Asian, and reporting high-risk behaviours, compared with HBV mono-infected patients. Cirrhosis, complications of end-stage liver disease, and liver transplantation were significantly more frequent in the HDV+ cohort. HDV viraemia was significantly associated with elevated liver transaminases and cirrhosis. Five HDV genotypes were observed among referred patients but no association between genotype and clinical outcome was detected within the HDV+ cohort.
Conclusions: Nearly 5% of the Canadian HBV referral population is HDV seropositive. HDV infection is highly associated with risk behaviours and both domestic and foreign-born patients with CHB. HDV was significantly associated with progressive liver disease highlighting the need for increased screening and surveillance of HDV in Canada.
Lay summary: Evidence of HDV infection was observed in approximately 5% of Canadians who were infected with HBV referred to medical specialists. HDV-positive patients were more likely to be male, born in Canada, or White or Black/African/Caribbean compared to Asian, and to have reported high-risk activities such as injection or intranasal drug use or high-risk sexual contact compared with patients infected with only HBV. Patients infected with HDV were also more likely to suffer severe liver disease, including liver cancer, compared with HBV mono-infected patients.
Keywords: ALT, alanine aminotransferase; AST, aspartate aminotransferase; CHB, chronic HBV infection; CanHepB Network, Canadian HBV network; Cirrhosis; Epidemiology; Genotype; HCC, hepatocellular carcinoma; Hepatitis B virus; Hepatitis D virus; IFNα, pegylated interferon-alpha; INR, international normalised ratio (prothrombin time of blood clotting); NAs, nucleos(t)ide analogue inhibitors; NML, National Microbiology Laboratory; TE, transient elastography; bp, base pairs; gt, genotype; n, count; qHBsAg, quantitative HBsAg.
Crown Copyright © 2022 Published by Elsevier B.V. on behalf of European Association for the Study of the Liver (EASL).
Conflict of interest statement
CSC within past 36 months: Grant or contract from Gilead Sciences, Janssen Pharmaceuticals, GSK; Consulting fees from Janssen Pharmaceuticals; Payment or honoraria from Gilead Sciences (all made to the University of Calgary). The other authors declare no conflicts of interest that pertain to this work. Please refer to the accompanying ICMJE disclosure forms for further details.
Figures


References
-
- Rizzetto M., Hamid S. The medical impact of hepatitis D virus infection in Asia and Africa; time for a reappraisal. Liver Int. 2021;41:16–19. - PubMed
-
- Alfaiate D., Clément S., Gomes D., Goossens N., Negro F. Chronic hepatitis D and hepatocellular carcinoma: a systematic review and meta-analysis of observational studies. J Hepatol. 2020;73:533–539. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

