Similarity and consistency assessment of three major online drug-drug interaction resources
- PMID: 35362214
- PMCID: PMC9545693
- DOI: 10.1111/bcp.15341
Similarity and consistency assessment of three major online drug-drug interaction resources
Abstract
Aims: The aim of this study was to explore the level of agreement on drug-drug interaction (DDI) information listed in three major online drug information resources (DIRs) in terms of: (1) interacting drug pairs; (2) severity rating; (3) evidence rating; and (4) clinical management recommendations.
Methods: We extracted information from the British National Formulary (BNF), Thesaurus and Micromedex. Following drug name normalisation, we estimated the overlap of the DIRs in terms of DDI. We annotated clinical management recommendations either manually, where possible, or through application of a machine learning algorithm.
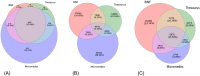
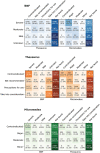
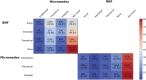
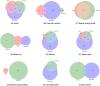
Results: The DIRs contained 51 481 (BNF), 38 037 (Thesaurus) and 65 446 (Micromedex) drug pairs involved in DDIs. The number of common DDIs across the three DIRs was 6970 (13.54% of BNF, 18.32% of Thesaurus and 10.65% of Micromedex). Micromedex and Thesaurus overall showed higher levels of similarity in their severity ratings, while the BNF agreed more with Micromedex on the critical severity ratings and with Thesaurus on the least significant ones. Evidence rating agreement between BNF and Micromedex was generally poor. Variation in clinical management recommendations was also identified, with some categories (i.e., Monitor and Adjust dose) showing higher levels of agreement compared to others (i.e., Use with caution, Wash-out, Modify administration).
Conclusions: There is considerable variation in the DDIs included in the examined DIRs, together with variability in categorisation of severity and clinical advice given. DDIs labelled as critical were more likely to appear in multiple DIRs. Such variability in information could have deleterious consequences for patient safety, and there is a need for harmonisation and standardisation.
Keywords: clinical decision support; clinical management of drug interactions; drug information; drug-drug interaction; drug-drug interaction software.
© 2022 The Authors. British Journal of Clinical Pharmacology published by John Wiley & Sons Ltd on behalf of British Pharmacological Society.
Conflict of interest statement
E.K. receives a PhD studentship that is jointly funded by AstraZeneca and the EPSRC. She also worked on a fixed‐term employment contract for AstraZeneca when this article was prepared. A.B. is an employee of Ceva Santé Animale. M.P. has received partnership funding for the following: MRC Clinical Pharmacology Training Scheme (co‐funded by MRC and Roche, UCB, Eli Lilly and Novartis); and grant funding from Vistagen Therapeutics. He also has unrestricted educational grant support for the UK Pharmacogenetics and Stratified Medicine Network from Bristol‐Myers Squibb and UCB. He has developed an HLA genotyping panel with MC Diagnostics, but does not benefit financially from this. M.P. is part of the IMI Consortium ARDAT (
Figures
Similar articles
-
The prevalence of major drug-drug interactions in older adults with cancer and the role of clinical decision support software.J Geriatr Oncol. 2018 Sep;9(5):526-533. doi: 10.1016/j.jgo.2018.02.001. Epub 2018 Apr 13. J Geriatr Oncol. 2018. PMID: 29510896
-
Drug interactions with cholinesterase inhibitors: an analysis of the French pharmacovigilance database and a comparison of two national drug formularies (Vidal, British National Formulary).Drug Saf. 2007;30(11):1063-71. doi: 10.2165/00002018-200730110-00005. Drug Saf. 2007. PMID: 17973542
-
Clinically significant drug-drug interactions between oral anticancer agents and nonanticancer agents: profiling and comparison of two drug compendia.Ann Pharmacother. 2008 Dec;42(12):1737-48. doi: 10.1345/aph.1L255. Epub 2008 Nov 25. Ann Pharmacother. 2008. PMID: 19033481 Review.
-
Comparison of potential drug-drug interactions with metabolic syndrome medications detected by two databases.PLoS One. 2019 Nov 14;14(11):e0225239. doi: 10.1371/journal.pone.0225239. eCollection 2019. PLoS One. 2019. PMID: 31725785 Free PMC article.
-
Drug-drug interactions in Neonatal Intensive Care Units: how to overcome a challenge.Minerva Pediatr (Torino). 2021 Apr;73(2):188-197. doi: 10.23736/S2724-5276.19.05388-X. Epub 2019 Oct 11. Minerva Pediatr (Torino). 2021. PMID: 31621271 Review.
Cited by
-
medicX-KG: a knowledge graph for pharmacists' drug information needs.J Biomed Semantics. 2025 Jul 1;16(1):11. doi: 10.1186/s13326-025-00332-7. J Biomed Semantics. 2025. PMID: 40597373 Free PMC article.
-
Clinical Relevance of Drug-Drug Interactions With Antibiotics as Listed in a National Medication Formulary: Results From Two Large Population-Based Case-Control Studies in Patients Aged 65-100 Years Using Linked English Primary Care and Hospital Data.Clin Pharmacol Ther. 2023 Feb;113(2):423-434. doi: 10.1002/cpt.2807. Epub 2022 Dec 16. Clin Pharmacol Ther. 2023. PMID: 36448824 Free PMC article.
-
Inconsistent Reporting of Interactions Between Warfarin and Medicinal Plants Across Clinical Decision Support Resources.J Evid Based Integr Med. 2025 Jan-Dec;30:2515690X251334445. doi: 10.1177/2515690X251334445. Epub 2025 Apr 21. J Evid Based Integr Med. 2025. PMID: 40255042 Free PMC article.
-
Analysis of queries to a Swedish drug information centre identifies scientific knowledge gaps.Sci Rep. 2024 Dec 6;14(1):30412. doi: 10.1038/s41598-024-82324-8. Sci Rep. 2024. PMID: 39638886 Free PMC article.
-
Drug-Drug-Gene Interactions in Cardiovascular Medicine.Pharmgenomics Pers Med. 2022 Nov 2;15:879-911. doi: 10.2147/PGPM.S338601. eCollection 2022. Pharmgenomics Pers Med. 2022. PMID: 36353710 Free PMC article. Review.
References
-
- Department of Health and Social Care . Good for You, Good for Us, Good for Everybody: A Plan to Reduce Overprescribing to Make Patient Care Better and Safer, Support the NHS, and Reduce Carbon Emissions; 2021. https://assets.publishing.service.gov.uk/government/uploads/system/uploa.... Published September 22, 2021. Accessed April 3, 2022.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

