Lipid-based strategies used to identify extracellular vesicles in flow cytometry can be confounded by lipoproteins: Evaluations of annexin V, lactadherin, and detergent lysis
- PMID: 35362259
- PMCID: PMC8971177
- DOI: 10.1002/jev2.12200
Lipid-based strategies used to identify extracellular vesicles in flow cytometry can be confounded by lipoproteins: Evaluations of annexin V, lactadherin, and detergent lysis
Abstract
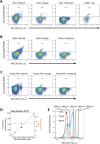
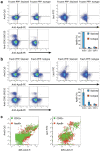
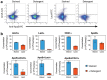
Flow cytometry (FCM) is a popular method used in characterisation of extracellular vesicles (EVs). Circulating EVs are often identified by FCM by exploiting the lipid nature of EVs by staining with Annexin V (Anx5) or lactadherin against the membrane phospholipid phosphatidylserine (PS) and evaluating the specificity of the labels by detergent lysis of EVs. Here, we investigate whether PS labelling and detergent lysis approaches are confounded by lipoproteins, another family of lipid-based nanoparticles found in blood, in both frozen and fresh blood plasma. We demonstrated that Anx5 and lactadherin in addition to EVs stained ApoB-containing lipoproteins, identified by the use of fluorophore-labelled polyclonal ApoB-antibody, and that Anx5 had a significantly larger tendency for labelling lipoprotein-bound PS than lactadherin. Furthermore, detergent lysis resulted in a decrease in both EV and lipoprotein events and especially lipoproteins positive for either Anx5 or lactadherin. Taken together, our findings pose concerns to the use of lipid-based strategies in identifying EVs by FCM and support the use of transmembrane proteins such as tetraspannins to distinguish EVs from lipoproteins.
Keywords: VLDL; annexin V; chylomicrons; detergent lysis; exosomes; extracellular vesicles; flow cytometry; lactadherin; lipids; lipoproteins; phosphatidylserine; triton X-100.
© 2022 The Authors. Journal of Extracellular Vesicles published by Wiley Periodicals, LLC on behalf of the International Society for Extracellular Vesicles.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Fluorescence triggering: A general strategy for enumerating and phenotyping extracellular vesicles by flow cytometry.Cytometry A. 2016 Feb;89(2):184-95. doi: 10.1002/cyto.a.22669. Epub 2015 Apr 9. Cytometry A. 2016. PMID: 25857288
-
A simple flow cytometry method improves the detection of phosphatidylserine-exposing extracellular vesicles.J Thromb Haemost. 2015 Feb;13(2):237-47. doi: 10.1111/jth.12767. Epub 2014 Nov 29. J Thromb Haemost. 2015. PMID: 25348269 Free PMC article.
-
Measurement of annexin V uptake and lactadherin labeling for the quantification of apoptosis in adherent Tca8113 and ACC-2 cells.Braz J Med Biol Res. 2008 Sep;41(9):750-7. doi: 10.1590/s0100-879x2008000900002. Braz J Med Biol Res. 2008. PMID: 18820763
-
A compendium of single extracellular vesicle flow cytometry.J Extracell Vesicles. 2023 Feb;12(2):e12299. doi: 10.1002/jev2.12299. J Extracell Vesicles. 2023. PMID: 36759917 Free PMC article. Review.
-
Lactadherin: An unappreciated haemostasis regulator and potential therapeutic agent.Vascul Pharmacol. 2018 Feb;101:21-28. doi: 10.1016/j.vph.2017.11.006. Epub 2017 Nov 21. Vascul Pharmacol. 2018. PMID: 29169950 Review.
Cited by
-
A New Strategy to Functionalize Exosomes via Enzymatic Engineering of Surface Glycans and its Application to Profile Exosomal Glycans and Endocytosis.Adv Sci (Weinh). 2025 May;12(18):e2415942. doi: 10.1002/advs.202415942. Epub 2025 Mar 19. Adv Sci (Weinh). 2025. PMID: 40106306 Free PMC article.
-
Expression of Tissue Factor and Platelet/Leukocyte Markers on Extracellular Vesicles Reflect Platelet-Leukocyte Interaction in Severe COVID-19.Int J Mol Sci. 2023 Nov 28;24(23):16886. doi: 10.3390/ijms242316886. Int J Mol Sci. 2023. PMID: 38069209 Free PMC article.
-
Phenotypic and proteomic analysis of plasma extracellular vesicles highlights them as potential biomarkers of primary Sjögren syndrome.Front Immunol. 2023 Jul 17;14:1207545. doi: 10.3389/fimmu.2023.1207545. eCollection 2023. Front Immunol. 2023. PMID: 37529039 Free PMC article.
-
The fluorochrome-to-protein ratio is crucial for the flow cytometric detection of tissue factor on extracellular vesicles.Sci Rep. 2024 Mar 17;14(1):6419. doi: 10.1038/s41598-024-56841-5. Sci Rep. 2024. PMID: 38494537 Free PMC article.
-
MIBlood-EV: Minimal information to enhance the quality and reproducibility of blood extracellular vesicle research.J Extracell Vesicles. 2023 Dec;12(12):e12385. doi: 10.1002/jev2.12385. J Extracell Vesicles. 2023. PMID: 38063210 Free PMC article.
References
-
- Aass, H. C. D. , Øvstebø, R. , Trøseid, A‐M. S. , Kierulf, P. , Berg, J. P. , & Henriksson, C. E. (2011). Fluorescent particles in the antibody solution result in false TF‐ and CD14‐positive microparticles in flow cytometric analysis. Cytometry Part A: The Journal of the International Society for Analytical Cytology, 79(12), 990–999. 10.1002/cyto.a.21147 - DOI - PubMed
-
- Andree, H. A. , Stuart, M. C. , Hermens, W. T. , Reutelingsperger, C. P. , Hemker, H. C. , Frederik, P. M. , & Willems, G. M. (1992). Clustering of lipid‐bound annexin V may explain its anticoagulant effect. The Journal of Biological Chemistry, 267(25), 17907–17912. 10.1016/s0021-9258(19)37128-5 - DOI - PubMed
-
- Arraud, N. , Gounou, C. , Turpin, D. , & Brisson, A. R. (2016). Fluorescence triggering: A general strategy for enumerating and phenotyping extracellular vesicles by flow cytometry. Cytometry Part A : The Journal of The International Society for Analytical Cytology, 89(2), 184–195. 10.1002/cyto.a.22669 - DOI - PubMed
-
- Arraud, N. , Linares, R. , Tan, S. , Gounou, C. , Pasquet, J. ‐ M. , Mornet, S. , & Brisson, A. R. (2014). Extracellular vesicles from blood plasma: Determination of their morphology, size, phenotype and concentration. Journal of thrombosis and haemostasis : JTH, 12(5), 614–627. 10.1111/jth.12554 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

