Spatial Distribution of Intracellular Ion Concentrations in Aggregate-Forming HeLa Cells Analyzed by μ-XRF Imaging
- PMID: 35363437
- PMCID: PMC8973254
- DOI: 10.1002/open.202200024
Spatial Distribution of Intracellular Ion Concentrations in Aggregate-Forming HeLa Cells Analyzed by μ-XRF Imaging
Abstract
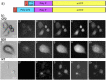
Protein aggregation is a hallmark of several severe neurodegenerative disorders such as Huntington's, Parkinson's, or Alzheimer's disease. Metal ions play a profound role in protein aggregation and altered metal-ion homeostasis is associated with disease progression. Here we utilize μ-X-ray fluorescence imaging in combination with rapid freezing to resolve the elemental distribution of phosphorus, sulfur, potassium, and zinc in huntingtin exon-1-mYFP expressing HeLa cells. Using quantitative XRF analysis, we find a threefold increase in zinc and a 10-fold enrichment of potassium that can be attributed to cellular stress response. While the averaged intracellular ion areal masses are significantly different in aggregate-containing cells, a local intracellular analysis shows no different ion content at the location of intracellular inclusion bodies. The results are compared to corresponding experiments on HeLa cells forming pseudoisocyanine chloride aggregates. As those show similar results, changes in ion concentrations are not exclusively linked to huntingtin exon-1 amyloid formation.
Keywords: X-ray fluorescence; huntingtin; potassium; pseudoisocyanine chloride; zinc.
© 2022 The Authors. Published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Hoffmann E. K., Lambert I. H., Pedersen S. F., Physiol. Rev. 2009, 89, 193–277. - PubMed
-
- Graham S. F., Nasarauddin M. B., Carey M., McGuinness B., Holscher C., Kehoe P. G., Love S., Passmore A. P., Elliott C. T., Meharg A., Green B. D., JAD 2015, 44, 851–857. - PubMed
-
- Kondratskyi A., Kondratska K., Skryma R., Prevarskaya N., Biochimica et Biophysica Acta (BBA) – Biomembranes 2015, 1848, 2532–2546. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

