Comprehensive genomics in androgen receptor-dependent castration-resistant prostate cancer identifies an adaptation pathway mediated by opioid receptor kappa 1
- PMID: 35365763
- PMCID: PMC8976065
- DOI: 10.1038/s42003-022-03227-w
Comprehensive genomics in androgen receptor-dependent castration-resistant prostate cancer identifies an adaptation pathway mediated by opioid receptor kappa 1
Erratum in
-
Author Correction: Comprehensive genomics in androgen receptor-dependent castration-resistant prostate cancer identifies an adaptation pathway mediated by opioid receptor kappa 1.Commun Biol. 2022 Jul 19;5(1):718. doi: 10.1038/s42003-022-03707-z. Commun Biol. 2022. PMID: 35854103 Free PMC article. No abstract available.
Abstract
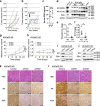
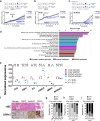
Castration resistance is a lethal form of treatment failure of prostate cancer (PCa) and is associated with ligand-independent activation of the androgen receptor (AR). It is only partially understood how the AR mediates survival and castration-resistant growth of PCa upon androgen deprivation. We investigated integrative genomics using a patient-derived xenograft model recapitulating acquired, AR-dependent castration-resistant PCa (CRPC). Sequencing of chromatin immunoprecipitation using an anti-AR antibody (AR-ChIP seq) revealed distinct profiles of AR binding site (ARBS) in androgen-dependent and castration-resistant xenograft tumors compared with those previously reported based on human PCa cells or tumor tissues. An integrative genetic analysis identified several AR-target genes associated with CRPC progression including OPRK1, which harbors ARBS and was upregulated upon androgen deprivation. Loss of function of OPRK1 retarded the acquisition of castration resistance and inhibited castration-resistant growth of PCa both in vitro and in vivo. Immunohistochemical analysis showed that expression of OPRK1, a G protein-coupled receptor, was upregulated in human prostate cancer tissues after preoperative androgen derivation or CRPC progression. These data suggest that OPRK1 is involved in post-castration survival and cellular adaptation process toward castration-resistant progression of PCa, accelerating the clinical implementation of ORPK1-targeting therapy in the management of this lethal disease.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J. Clin. 2020;70:7–30. - PubMed
-
- Hahn AW, Higano CS, Taplin ME, Ryan CJ, Agarwal N. Metastatic castration-sensitive prostate cancer: optimizing patient selection and treatment. Am. Soc. Clin. Oncol. Educ. Book. 2018;38:363–371. - PubMed
-
- Tombal B. What is the pathophysiology of a hormone-resistant prostate tumour? Eur. J. Cancer. 2011;47:S179–S188. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

