Proton Quantum Tunneling: Influence and Relevance to Acidosis-Induced Cardiac Arrhythmias/Cardiac Arrest
- PMID: 35366283
- PMCID: PMC8830476
- DOI: 10.3390/pathophysiology28030027
Proton Quantum Tunneling: Influence and Relevance to Acidosis-Induced Cardiac Arrhythmias/Cardiac Arrest
Abstract
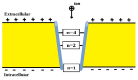
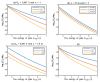
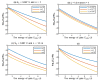
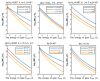
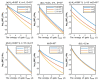
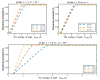
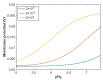
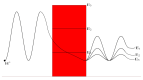
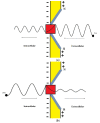
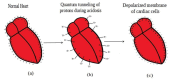
Acidosis and its associated pathologies predispose patients to develop cardiac arrhythmias and even cardiac arrest. These arrhythmias are assumed to be the result of membrane depolarization, however, the exact mechanism of depolarization during acidosis is not well defined. In our study, the model of quantum tunneling of protons is used to explain the membrane depolarization that occurs during acidosis. It is found that protons can tunnel through closed activation and inactivation gates of voltage-gated sodium channels Nav1.5 that are present in the membrane of cardiac cells. The quantum tunneling of protons results in quantum conductance, which is evaluated to assess its effect on membrane potential. The quantum conductance of extracellular protons is higher than that of intracellular protons. This predicts an inward quantum current of protons through the closed sodium channels. Additionally, the values of quantum conductance are influential and can depolarize the membrane potential according to the quantum version of the GHK equation. The quantum mechanism of depolarization is distinct from other mechanisms because the quantum model suggests that protons can directly depolarize the membrane potential, and not only through indirect effects as proposed by other mechanisms in the literature. Understanding the pathophysiology of arrhythmias mediated by depolarization during acidosis is crucial to treat and control them and to improve the overall clinical outcomes of patients.
Keywords: acidosis; arrhythmias; proton; quantum biology; quantum conductance; quantum tunneling; voltage-gated channels.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Greenberg A. Primer on Kidney Diseases E-Book. Elsevier Health Sciences; Amsterdam, The Netherlands: 2009.
-
- Hall J.E., Hall M.E. Guyton and Hall Textbook of Medical Physiology E-Book. Elsevier Health Sciences; Amsterdam, The Netherlands: 2020.
-
- Gerst P.H., Fleming W.H., Malm J.R. Increased susceptibility of the heart to ventricular fibrillation during metabolic acidosis. Circ. Res. 1966;19:63–70. doi: 10.1161/01.RES.19.1.63. - DOI
LinkOut - more resources
Full Text Sources
