Vascular Injury in the Zebrafish Tail Modulates Blood Flow and Peak Wall Shear Stress to Restore Embryonic Circular Network
- PMID: 35369301
- PMCID: PMC8971683
- DOI: 10.3389/fcvm.2022.841101
Vascular Injury in the Zebrafish Tail Modulates Blood Flow and Peak Wall Shear Stress to Restore Embryonic Circular Network
Abstract
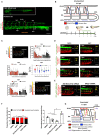
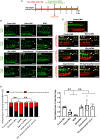
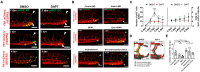
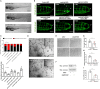
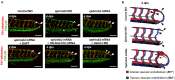
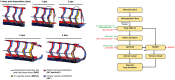
Mechano-responsive signaling pathways enable blood vessels within a connected network to structurally adapt to partition of blood flow between organ systems. Wall shear stress (WSS) modulates endothelial cell proliferation and arteriovenous specification. Here, we study vascular regeneration in a zebrafish model by using tail amputation to disrupt the embryonic circulatory loop (ECL) at 3 days post fertilization (dpf). We observed a local increase in blood flow and peak WSS in the Segmental Artery (SeA) immediately adjacent to the amputation site. By manipulating blood flow and WSS via changes in blood viscosity and myocardial contractility, we show that the angiogenic Notch-ephrinb2 cascade is hemodynamically activated in the SeA to guide arteriogenesis and network reconnection. Taken together, ECL amputation induces changes in microvascular topology to partition blood flow and increase WSS-mediated Notch-ephrinb2 pathway, promoting new vascular arterial loop formation and restoring microcirculation.
Keywords: Notch-ephrinb2 signaling; biophysics; peak wall shear stress; vascular injury and repair; vascular loop formation.
Copyright © 2022 Baek, Chang, Chang, Roustaei, Ding, Wang, Chen, O'Donnell, Chen, Ashby, Xu, Mack, Cavallero, Roper and Hsiai.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Computational simulations of the 4D micro-circulatory network in zebrafish tail amputation and regeneration.J R Soc Interface. 2022 Feb;19(187):20210898. doi: 10.1098/rsif.2021.0898. Epub 2022 Feb 16. J R Soc Interface. 2022. PMID: 35167770 Free PMC article.
-
High-Throughput Imaging of Blood Flow Reveals Developmental Changes in Distribution Patterns of Hemodynamic Quantities in Developing Zebrafish.Front Physiol. 2022 Jun 20;13:881929. doi: 10.3389/fphys.2022.881929. eCollection 2022. Front Physiol. 2022. PMID: 35795647 Free PMC article.
-
Endothelial Shc regulates arteriogenesis through dual control of arterial specification and inflammation via the notch and nuclear factor-κ-light-chain-enhancer of activated B-cell pathways.Circ Res. 2013 Jun 21;113(1):32-39. doi: 10.1161/CIRCRESAHA.113.301407. Epub 2013 May 9. Circ Res. 2013. PMID: 23661718 Free PMC article.
-
Regulation of coronary blood flow during exercise.Physiol Rev. 2008 Jul;88(3):1009-86. doi: 10.1152/physrev.00045.2006. Physiol Rev. 2008. PMID: 18626066 Review.
-
Fluid Shear Stress Sensing by the Endothelial Layer.Front Physiol. 2020 Jul 24;11:861. doi: 10.3389/fphys.2020.00861. eCollection 2020. Front Physiol. 2020. PMID: 32848833 Free PMC article. Review.
Cited by
-
Aortopathy associated with bicuspid aortic valve: advances in clinical and hemodynamics research.Front Physiol. 2025 May 6;16:1576072. doi: 10.3389/fphys.2025.1576072. eCollection 2025. Front Physiol. 2025. PMID: 40395642 Free PMC article. Review.
-
Flow-induced reprogramming of endothelial cells in atherosclerosis.Nat Rev Cardiol. 2023 Nov;20(11):738-753. doi: 10.1038/s41569-023-00883-1. Epub 2023 May 24. Nat Rev Cardiol. 2023. PMID: 37225873 Free PMC article. Review.
-
Application of a ImageJ-Based Method to Measure Blood Flow in Adult Zebrafish and Its Applications for Toxicological and Pharmacological Assessments.Biology (Basel). 2025 Jan 10;14(1):51. doi: 10.3390/biology14010051. Biology (Basel). 2025. PMID: 39857282 Free PMC article.
-
Mapping the cellular expression patterns of vascular endothelial growth factor aa and bb genes and their receptors in the adult zebrafish brain during constitutive and regenerative neurogenesis.Neural Dev. 2024 Sep 12;19(1):17. doi: 10.1186/s13064-024-00195-1. Neural Dev. 2024. PMID: 39267104 Free PMC article.
-
Hemodynamic regulation allows stable growth of microvascular networks.Proc Natl Acad Sci U S A. 2024 Feb 27;121(9):e2310993121. doi: 10.1073/pnas.2310993121. Epub 2024 Feb 22. Proc Natl Acad Sci U S A. 2024. PMID: 38386707 Free PMC article.
References
-
- Tanaka H, Shimizu S, Ohmori F, Muraoka Y, Kumagai M, Yoshizawa M, et al. . Increases in blood flow and shear stress to nonworking limbs during incremental exercise. Med Sci Sports Exerc. (2006) 38:81–5. 10.1249/01.mss.0000191166.81789.de - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

